It has been some 18 months since my last posts on autism
An update on new studies since then:
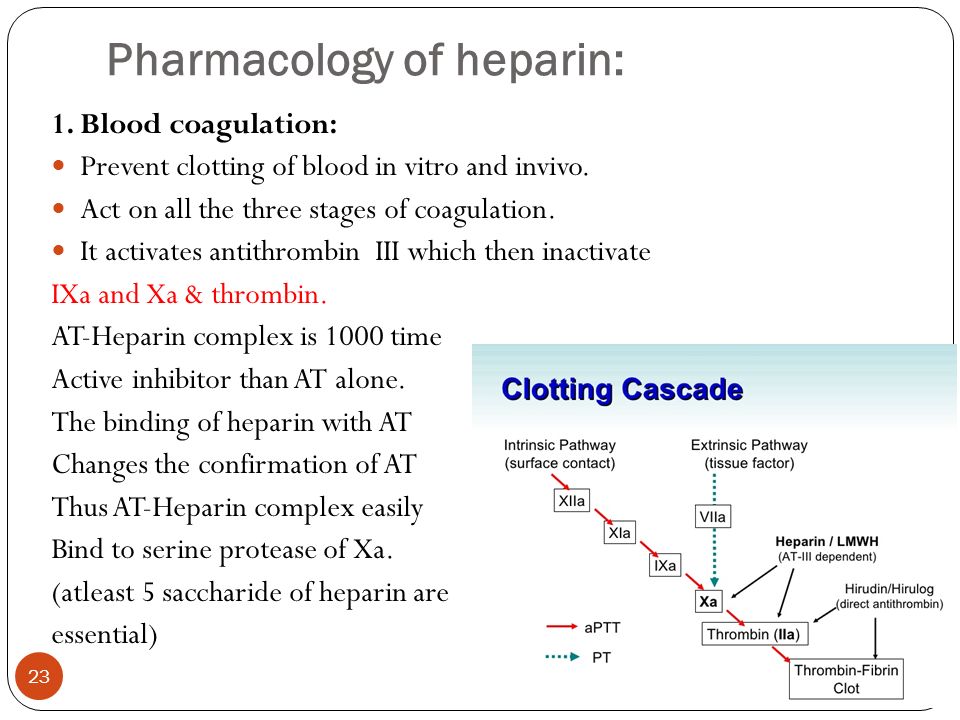
- ” we found an increased hazard ratio of autism associated with otitis media[inflammation of the middle ear] (1.83 95% CI 1.71-1.95) and antibiotics usage (1.29 95% CI 1.17-1.43). ” [2018]
- “In the sibling-stratified Cox model, only broader spectrum antibiotics were associated with increased risk of autism: hazard ratio (HR) = 1.16 ” [2019]
- ” Among 3253 children with ASD, 37.0% had a GI-related diagnosis during the last 2 years of their 5-year health coverage enrollment period, compared to 20.0% of 278,370 children from the general population without an ASD diagnosis. Greater numbers of oral antibiotic fills during the first 3 years of enrollment were found to significantly increase the hazard rate of having a later GI-related diagnosis (adjusted hazard ratio 1.48; 95% confidence interval 1.34, 1.63) in children both with and without ASD. ” [2018]
- Alteration of gut microbiota-associated epitopes in children with autism spectrum disorders[2019].
- The Gut Microbiota and Dysbiosis in Autism Spectrum Disorders [2018].
- The role of microbiota in autism spectrum disorders[2018].
- Gut Microbial Dysbiosis in Indian Children with Autism Spectrum Disorders [2018].
- ” we demonstrate a molecular basis for how the host microbiome is crucial for a normal behavioural response during social interaction. Our data further suggest that social behaviour is correlated with the gene-expression response in the amygdala, established during neurodevelopment as a result of host-microbe interactions. ” [2018]
- ” In this review, the latest evidence linking mitochondrial dysfunction and abnormalities in mitochondrial DNA (mtDNA) to the pathogenesis of autism will be presented. ” [2018] – Ouch, it’s associated with the mother’s DNA, or mutations from it.
- ” we uncovered elevated common polygenic risk, …and a maternally inherited X-linked deletion ” [2018]
- “in the results found in this review, the highest prevalence of the C. albicans in children with ASD stands out. However, little is still known about the involvement of Candida spp., and other types of fungi, on gastrointestinal symptoms and ASD symptoms, in children with ASD. ” [2019]
- Anti-Candida albicans IgG Antibodies in Children With Autism Spectrum Disorders[2018].
- “ASD children had a higher rate of high-positive values compared to typically developed children with an unadjusted odds ratio of 3.45”
- Reduced microbiome alpha diversity in young patients with ADHD. [2018]
- The valproic acid rat model of autism presents with gut bacterial dysbiosis similar to that in human autism [2018].
- ” In conclusion, our data on the gut microbial community of the 400-E12 rats in response to prenatal VPA exposure indicate that this model, in addition to demonstrating behavioral and anatomical similarities to autism, also mimics the microbiota features of autism, making it one of the best-suited rodent models for the study of autism. “
- Alteration of gut microbiota-associated epitopes in children with autism spectrum disorders[2019].
- ” In this review, the latest evidence linking mitochondrial dysfunction and abnormalities in mitochondrial DNA (mtDNA) to the pathogenesis of autism will be presented. ” [2018] – Ouch, it’s associated with the mother’s DNA, or mutations from it.
- ” we uncovered elevated common polygenic risk, …and a maternally inherited X-linked deletion ” [2018]
- “in the results found in this review, the highest prevalence of the C. albicans in children with ASD stands out. However, little is still known about the involvement of Candida spp., and other types of fungi, on gastrointestinal symptoms and ASD symptoms, in children with ASD. ” [2019]
- Anti-Candida albicans IgG Antibodies in Children With Autism Spectrum Disorders[2018].
- “ASD children had a higher rate of high-positive values compared to typically developed children with an unadjusted odds ratio of 3.45”
- “At the genus level, we found that the relative abundance of Sutterella, Odoribacter and Butyricimonas was much more abundant in the ASD group whereas the abundance of Veillonella and Streptococcus was decreased significantly compared to the control group. ” [2018]
- The valproic acid rat model of autism presents with gut bacterial dysbiosis similar to that in human autism [2018].
- ” In conclusion, our data on the gut microbial community of the 400-E12 rats in response to prenatal VPA exposure indicate that this model, in addition to demonstrating behavioral and anatomical similarities to autism, also mimics the microbiota features of autism, making it one of the best-suited rodent models for the study of autism. “

Treatment Implications
- Diet Can Impact Microbiota Composition in Children With Autism Spectrum Disorder.[2018]
- Probiotic treatment reduces the autistic-like excitation/inhibition imbalance in juvenile hamsters induced by orally administered propionic acid and clindamycin [2018].
” we found that treatment with L. reuteri selectively rescues social deficits in genetic, environmental, and idiopathic ASD models. Interestingly, the effects of L. reuteri on social behavior are not mediated by restoring the composition of the host’s gut microbiome, which is altered in all of these ASD models. Instead, L. reuteri acts in a vagus nerve-dependent manner and rescues social interaction-induced synaptic plasticity in the ventral tegmental area of ASD mice, but not in oxytocin receptor-deficient mice. Collectively, treatment with L. reuteri emerges as promising non-invasive microbial-based avenue to combat ASD-related social dysfunction. ”
Mechanisms Underlying Microbial-Mediated Changes in Social Behavior in Mouse Models of Autism Spectrum Disorder.
There is promising evidence to suggest that probiotic therapy may improve gastrointestinal dysfunction, beneficially alter fecal microbiota, and reduce the severity of ASD symptoms in children with ASD. Future research is still warranted in this area because there are methodologic flaws in the available literature and optimal species, strains, dosages, and duration of treatment have not been identified.
Role of Probiotics in Managing Gastrointestinal Dysfunction in Children with Autism Spectrum Disorder: An Update for Practitioners. [2018]
The results showed that children on exclusion diets reported significantly lower scores of abdominal pain and bowel movement, as well as lower abundance of Bifidobacterium spp. and Veillonellaceae family, but higher presence of Faecalibacterium prausnitzii and Bacteroides spp. In addition, significant correlations were found between bacterial populations and faecal amino acids in this group, compared to children following an unrestricted diet. Following B-GOS® intervention, we observed improvements in anti-social behaviour, significant increase of Lachnospiraceae family, and significant changes in faecal and urine metabolites.
A prebiotic intervention study in children with autism spectrum disorders (ASDs). [2018]
Bottom Line
Autism appears to be associated with two major factors: mutations in the DNA, especially in mtDNA. and microbiome. DNA and the microbiome have intense interplay with microbiome shifts likely causing epigenetic changes in DNA in some cases.
Altering the microbiome (although complex to do), promises the greatest hope in moderating the symptoms of Autism and ASD.
I have updated my microbiome analysis site with additional information taken from the various articles cited above for those who have had microbiome done by ubiome.com or others.

For those that have not, there is a Apriori Adjustment page located here.