Recently I visited my dentist and during the visit, she and me got into an interesting discussion of the microbiome and dentistry. She show me in my x-rays some interesting aspects of bacteria in the mouth.
“There seems to be an increased prevalence of periodontal disease in patients with IBD when compared to healthy controls, probably due to changes in the oral microbiota and a higher inflammatory response. Moreover, the induction of periodontitis seems to result in gut dysbiosis and altered gut epithelial cell barrier function, which might contribute to the pathogenesis of IBD. Considering the complexity of both periodontal disease and IBD, it is very challenging to understand the possible pathways involved in their coexistence.” [2016]
Amalgam Impacts – Allergy
My dentist cited growing concerns about interactions between different metals being used for restorative work (i.e. electrolysis). Concerns included shifts of bacteria populations. Some literature:
- “three subjects with CFS and two with fibromyalgia, all of whom suspected metal exposure as a trigger for their ill health…All patients except one were sensitized to metals present in their dental restorations… The removal of sensitizing metals resulted in long-term health improvement.” [2014]
- “Fifteen female FM patients…Reduction of metal exposure in the FM patients was achieved by replacement of dental metal restorations and by the avoidance of known sources of metal exposure…. Objective examination 5 years later showed that half of the patients no longer fulfilled the FM diagnosis, 20% had improved and the remaining 30% still had FM. All patients reported subjective health improvement. This correlated with the normalization of metal-specific responses in vitro.” [2013]
- ” 15 patients who suffered from clinical metal sensitivity in addition to other health problems. … The removal of incompatible dental material (RID) resulted in long-term health improvement in the majority of patients. We postulate that in vivo, metal ions activate T-cells, initiating systemic inflammation, which, through cytokines, affects the brain and hypothalamus-pituitary-adrenal axis.” [2006]
- “Thirty-eight patients; 9 with systemic lupus erythematosus (SLE), 16 with rheumatoid arthritis (RA), and 13 with Sjögren’s syndrome (SS) and a control group of 43 healthy age- and sex-matched subjects were included in the study… The majority of patients (87%) had a positive lymphocyte reaction to at least one metal and 63% reacted to two or more metals tested…The most frequent allergens were nickel, mercury, gold and palladium.” [2015]
- ” Studies have shown that chronic Hg exposure from various sources including dental amalgams is associated with numerous health complaints, including fatigue, anxiety, and depression–and these are among the main symptoms that are associated with CFS and FM…several studies have shown that the removal of amalgams is associated with improvement in these symptoms.” [2014]
- “To evaluate clinical relevance of positive in vitro findings, the replacement of amalgam with metal-free restorations was performed in some of the patients. At a six-month follow-up, patients reported considerably alleviated fatigue and disappearance of many symptoms previously encountered; in parallel, lymphocyte responses to metals decreased as well. We suggest that metal-driven inflammation may affect the hypothalamic-pituitary-adrenal axis (HPA axis) and indirectly trigger psychosomatic multisymptoms characterizing chronic fatigue syndrome, fibromyalgia, and other diseases of unknown etiology.” [1999]
Metals and Microbiome
Assuming mercury alone is the problem and not the other base metals cited above, is likely false. Mercury is an easy target loaded with emotion.
- “The role of nickel in the biochemistry of bacterial enzymes is discussed here, covering both the beneficial effects of nickel in the human microbiota as well as the role of nickel-depending bacteria in human pathogenesis. In addition, the hazardous health effects caused by nickel exposure to humans, namely nickel-induced carcinogenesis and allergy, are triggered by non-specific interactions of nickel with macromolecules and formation of reactive compounds that mediate cellular damage. Cellular response to nickel is also related to signal transduction cascades.” [2016]
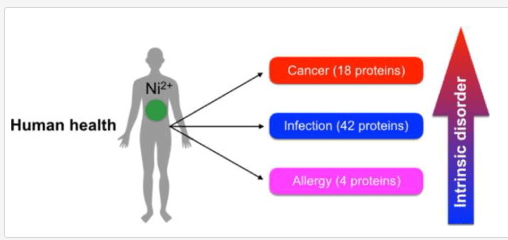
- “The bacterial count results showed that dietary nickel (NiCl2) in the range of 300 to 900 mg/kg decreased the counts of Bifidobacterium spp. and Lactobacillus, increased Escherichia coli (E. coli) and Enterococcus spp. in the ileum and cecum. …This finding implies that NiCl2 has toxicological effect on the intestinal ecosystem and, possibly functions.” [2014]
Root Canals/Dentures as bacteria reserves
I view the mouth as a reserve for many bacteria that contribute to CFS/FM/IBS — fixing the guts may be temporary because the mouth may repopulate it with the bad bacteria.
- Exploration of bacterial species associated with the salivary microbiome of individuals with a low susceptibility to dental caries[2016]. “These results revealed an association between a greater abundance of Porphyromonas pasteri and lower susceptibility to dental caries.”
- “IMPORTANCE Denture stomatitis is a prevalent inflammatory condition of the mucosal tissue in denture wearers that is triggered by microorganisms. While Candida has been extensively studied for its role in stomatitis etiology, the bacterial component largely remains to be investigated. Our data show that certain types of bacteria are significantly associated with denture health and disease.” [2016]
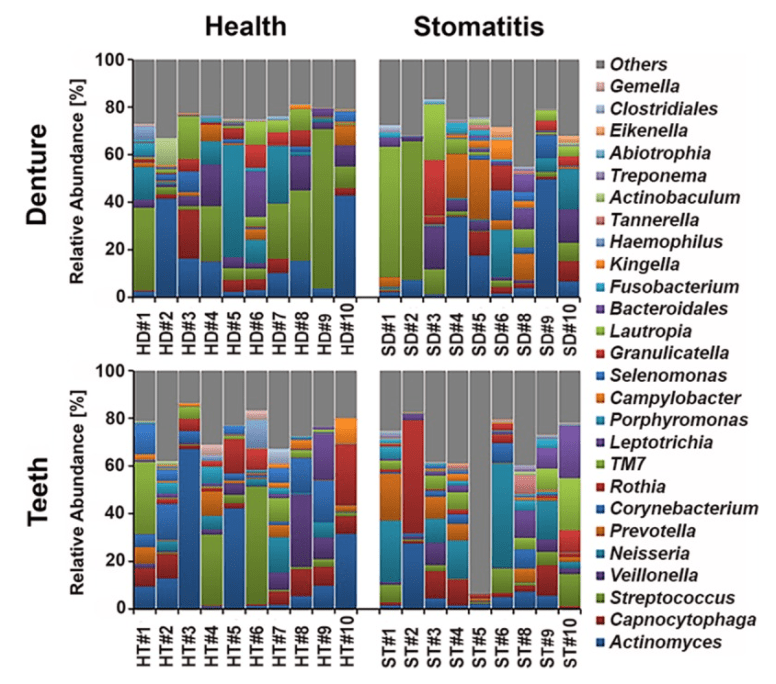
- As shown above — individuals vary greatly — there is no “normal” pattern.
Species to watch out for in microbiome results
- “evaluated the presence and relative levels of 28 bacterial taxa in treated root canals of teeth with posttreatment apical periodontitis … Taxa detected more frequently included Streptococcus species (47%), Lactobacillus species (35%), Dialister invisus (29%), Eubacterium infirmum (29%), Prevotella intermedia (29%), Selenomonas sputigena (29%), Synergistes oral clone BA121 (29%), and Treponema denticola (29%).” [2008]
- “Enterococcus faecalis was the most prevalent species, detected in 77.8% of the study teeth, followed by Peptostreptococcus micros, detected in 51.1%. Porphyromonas gingivalis, Porphyromonas endodontalis, Prevotella intermedia, and Prevotella nigrescens were detected in 35.6%, 22.2%, 11.1%, and 11.1% of the sampled teeth, respectively. Moreover, PCR detected Filifactor alocis in 26.7%, Treponema denticola in 24.4%, and Tannerella forsythia in 4.4% of the samples. [2008]
- T. denticola and P. micros were statistically associated with tenderness to percussion (p < 0.05).
- P. nigrescens was associated with the presence of spontaneous pain and abscess (p < 0.05).
- P. endodontalis and P. nigrescens were associated with purulent exudates (p < 0.05).
From Endotontic microbiology [2010] – longer lists are in this article.
1) Black pigmented Gram negative anaerobic rods include species formerly known as Bacteroides melaninogenicus. These bacteria have been reclassified into two genera: (a) saccharolytic species – Prevotella and (b) asaccharolytic species – Porphyromonas.[7]
Prevotella species detected in endodontic infections include
-
Prevotella intermedia
-
Prevotella nigrescens
-
Prevotella tannerae
-
Prevotella multissacharivorax
-
Prevotella baroniae and
-
Prevotella denticola.
Porphyromonas species detected in endodontic infections include
-
Porphyromonas endodontalis and
-
Porphyromonas gingivalis.
2) Tannerella forsythia (previously called Bacteroides forsythus or Tannerella forsythenis) was the first periodontal pathogen to be detected in endodontic infection.[8]
3) Dialister species are asaccharolytic obligately anaerobic Gram negative coccobacilli which have been consistently detected in endodontic infections.
-
Dialister pneumosintes and
-
Dialister invisus.
4) Fusobacterium is also a common member of endodontic microbiota.
-
Fusobacterium nucleatum
-
Fusobacterium periodonticum
5) Spirochetes are highly motile, spiral-shaped, Gram negative bacteria with periplasmic flagella. All oral spirochetes fall into the genus Treponema.[9] Prevalent species are
-
Treponema denticola
-
Treponema sacranskii
-
Treponema parvum
-
Treponema maltophilum and
-
Treponema lecithinolyticum.
6) Gram positive anaerobic rods have also been found in endodontic microbiota like
-
Pseudoramibacter alactolyticus
-
Filifactor alocis
-
Actinomyces spp.
-
Propionibacterium propionicum
-
Olsenella spp.
-
Slackia exigua
-
Mogibacterium timidum and
-
Eubacterium spp.
7) Gram positive cocci that are present in endodontic infection:
-
Parvimonas micra (previously called Peptostreptococcus micros or Micromonas micros)
-
Streptococcus spp. which include,Streptococcus anginosusStreptococcus mitisiStreptococcus sanguinis
-
Enterococcus faecalis.
Other bacterial spp. which are present in low to moderate values include
-
Campylobacter spp. which are Gram negative anaerobic rods; common species are,Campylobacter rectus andCampylobacter gracilis.
-
Catonella morbic which is a saccharolytic obligate anaerobic Gram negative rod
-
Veillonella parvula
-
Eikenella corrodens
-
Granulicatella adiacens
-
Neisseria mucosa
-
Centipeda periodontii
-
Gemella morbillorum
-
Capnocytophaga gingivalis
-
Corynebacterium matruchotii
-
Bifidobacterium dentium and
-
anaerobic lactobacilli.
If any of those show up, you may wish to pay special attention to your teeth.
Bottom Line
For some past restorative work may be a significant factor for FM/IBS/CFS, with all ceramic replacement being the best alternatives. Good dental health (i.e. no gum disease) may be essential for remission and maintaining remission.
