For the technical minded, I have just add a partial list of the articles that I am using.
http://microbiomeprescription.com/Library/Citations
Category Archives: Conditions
A possible subset of CFS with a smelly symptom
This week I got an email from activist in another under served medical condition, she wrote ” Preliminary microbiome results are most similar to CFS. “. In another sense, it’s may be a variation of SIBO. There is a DNA component (as there is with CFS, FM, IBS).
A list of common symptoms include:
- Fatigue
- Anxiety
- Especially temperature sensitivity,
- Presence sensitivity, “sometimes all it takes is another person coming next to me before I get extremely hot and a prickly sensation comes over my body, this usually comes with the odor getting worse for a few moments and it’s not related to anxiety as once I get these symptoms I’ll look around and then realize someone is there”
- General malaise
- Randomly getting very tired
- Recurrent sore throat
- Bruxism
- Difficulty processing information at times.
Some web links
- https://www.biorxiv.org/content/early/2017/05/19/139014
- “Most cases of socially-disabling idiopathic malodor remain undiagnosed and there are no guidelines for diagnostic tests nor treatment options that extend beyond TMAU”
- https://www.meboresearch.org/
- “MEBO’s Mission is to initiate research into rare genetic metabolic diseases, particularly those resulting in systemic, difficult to control body malodor and halitosis, including, but not limited to, Trimethylaminuria. “
- http://www.meboblog.com/
- https://allofusresearchpriorities.ideascale.com/a/dtd/Could-the-Precision-Medicine-Initiative-Reduce-Social-Inequality/195120-44880#idea-tab-comments
- https://systemicbodyodor.blogspot.com/
Despite being known for 3000 years, still little understanding. Trimethylamine is common in most food. it can be created by bacteria from almost any food, especially rich in protein; indoles (foods such as Brussel sprouts) inhibit enzyme “deodoryzing” trimethylamine in our liver. And, worst of all, almost nothing is known about trimethylaminuria-negative cases
PubMed
- “CLINICAL CHARACTERISTICS:Primary trimethylaminuria is characterized by a fishy odor resembling that of rotten or decaying fish that results from excess excretion of trimethylamine in the urine, breath, sweat, and reproductive fluids. No physical symptoms are associated with trimethylaminuria. Affected individuals appear normal and healthy; however, the unpleasant odor often results in social and psychological problems. Symptoms are usually present from birth and may worsen during puberty. In females, symptoms are more severe just before and during menstruation, after taking oral contraceptives, and around the time of menopause. [2015]
- “The fish malodor syndrome (also known as the fish odor syndrome and trimethylaminuria) is a metabolic disorder characterized by the presence of abnormal amounts of the dietary-derived tertiary amine, trimethylamine, in the urine, sweat, expired air, and other bodily secretions. Trimethylamine itself has the powerful aroma of rotting fish, and this confers upon the sufferer a highly objectionable body odor, which can be destructive to the personal, social, and work life of the affected individual… known since 1000BC from Mahabharata (Indian Epic)” [2001]
- “George Preti has been working on it for 30+ years, but as his latest paper showed they can’t connect even the most severe case of TMAU to one gene” from reader
- ” Our studies include patients who have been referred to us after being examined by numerous clinical specialists with no identification or relief from their problem. This is due in part to the intermittent nature of many of these problems as well as an apparent lack of knowledge concerning many of these metabolic problems and their relation to oral symptoms.” [1992]
- “Individuals with the metabolic disorder trimethylaminuria may sporadically produce malodors despite good hygiene. The psychosocial impact of trimethylaminuria can be considerable. However, trimethylaminuria is difficult to diagnose without specialized tests, in part because odor production is diet-dependent, and malodors may not be present during medical examinations. Thus, the prevalence and demographics of trimethylaminuria remain unclear… Consistent with previous reports, women, particularly African American women, were significantly overrepresented among trimethylaminuria-positive patients.” [2011] – Same situation as with CFS/FM — more women than men.
- “not necessarily – our pilot programs had more men, men are more likely to take risks, pay for experimental tests and test new treatments” reader
Clinical Studies
- https://clinicaltrials.gov/ct2/show/NCT03451994
- Exploratory Study of Volatile Organic Compounds in Alveolar Breath (VOCs)
- https://clinicaltrials.gov/ct2/show/NCT02692495
- Evaluation of Potential Screening Tools for Metabolic Body Odor and Halitosis
- https://clinicaltrials.gov/ct2/show/NCT02683876
-
Exploratory Study of Relationships Between Malodor and Urine Metabolomics
-
Current Treatment
“Treatment of manifestations: Dietary restriction of: Trimethylamine (present in milk obtained from wheat-fed cows) and its precursors including choline (present in eggs, liver, kidney, peas, beans, peanuts, soya products, and brassicas [Brussels sprouts, broccoli, cabbage, cauliflower]), lecithin and lecithin-containing fish oil supplements; Trimethylamine N-oxide (present in seafood [fish, cephalopods, and crustaceans]); Inhibitors of FMO3 enzyme activity such as indoles (found in brassicas). Use of: Acid soaps and body lotions to remove secreted trimethylamine by washing; Activated charcoal and copper chlorophyllin to sequester trimethylamine produced in the gut; Antibiotics (metronidazole, amoxicillin, and neomycin) to suppress production of trimethylamine by reducing bacteria in the gut; Riboflavin supplements to enhance residual FMO3 enzyme activity. Prevention of secondary complications: Planning and monitoring of diet to ensure that the daily intake of choline and folate meets recommendations for age and sex; no restriction of dietary choline during pregnancy and lactation. Agents/circumstances to avoid: Foods with a high content of precursors of trimethylamine or inhibitors of FMO3 enzyme activity (seafoods: fish, cephalopods, and crustaceans), eggs, offal, legumes, brassicas, and soya products; food supplements and “health” foods that contain high doses of choline and lecithin; drugs metabolized by the FMO3 enzyme; circumstances that promote sweating (exercise, stress, and emotional upsets). ” [2015]
- “Dr. Shephard now admits dietary restrictions are even more complicated – other food compounds that should be avoided are Ergothioneine, betaine. And the network of microbes messing up with it is quite large ” – reader
New Symptoms Added for this subset
The following overlap in some cases, and do not in others
- Comorbid: Malodor
- Any type of odor problem of uncertain origin
- Comorbid: Trimethylaminuria
- Ideally with a positive test
- Comorbid: Proximity triggered Malodor
- Comorbid: Chronic Halitosis
- A differentiation (see [2007])
- Autonomic Manifestations: Chemical Smelling Urine (atypical)
If the CFS diagnosis masked reporting malodor, please update your symptoms.
For new Malodor readers with uBiome
- Upload your data at http://microbiomeprescription.com/Home/Upload
- Recommendations on normalizing gut bacteria in general is given.
- In time, there may be customization to lower malodor associated bacteria added (need data first).
A post from a reader on microbiome mechanism
Ben posted this on an earlier post, I thought that it should be presented as a reader post.
This article may be of interest:
The researchers identified a new mechanism where repeated exposure to typical food poisoning pathogens lead to a chronic disease state that did not resolve by itself, even after the pathogens were cleared, and even if the levels of pathogen were less than is needed to produce acute food poisoning symptoms (so the hosts may not have been aware).
The mechanism is to do with an acquired deficiency of an important gut inflammation mediating enzyme – called Intestinal Alkaline Phosphatase ( IAP)
Without this enzyme, the host is unable to deal with normal bacterial toxins from even beneficial microbes in the gut, and a cascade of damaging inflammation is created in a self-perpetuating cycle.
- This leads to a big list of symptoms that are a pretty good match to much of what is described in CFS
- Inflammation of Colon and Small Intestine
- Leaky Gut (small intestine membrane permeability)
- Weight loss (likely indicates malabsorption)
- diahrea (or altered bowel habit)
- faecal blood – and prolapsed anus
- colon shortening
- systemic inflammation (host of inflammatory cytokines are increased by up to 26 fold)
- It’s pretty easy to see this might lead to further symptoms in a chain of events
- disrupted metabolism (due to systemic inflammation, oxidation etc )
- fatigue ( due to malabsorption, leaky gut or systemic inflammation etc)
- allergy / food sensitivity ( due to gut permeability)
- systemic cell aging – neuraminidases are known to regulate the aging of various enzymes and cells in the body
- hormonal / sympathetic nervous system disorders – again from systemic inflammation (dysregulated sleep, blood pressure, temperature, mood, etc
- neurological disorders – again from inflammation, dysbiois (memory, cognitive function, etc etc )
- clotting disorders – Neuraminidases are known to interfere with both cell membrane and clotting mechanisms
I mention it here as it’s a possible explanation or underlying model describing the experiences discussed here.
The fit seems very good.
Interestingly the condition also causes the host to lose control of the gut microbiota population resulting in imbalance in the proportions of beneficial and pathogenic strains. Further re-enforcing the cycle – and would match many of the gut symptoms reported by sufferers. And echo the Ubiome reports of people here.
Neuraminidases are also known to cause disruption of the clotting mechanisms in humans – so this could explain the link to red blood cell morphology in CFS sufferers and sticky blood as discussed here.
It would also explain the prolonged ‘Herx’ reactions that many sufferers report. When the mainstream view is that any true Herxheimer reaction lasts only a few days as microbes in the blood are killed. But, if an overactive immune response to the LPS toxins of microbes in the gut is responsible then we would indeed expect a long-term reaction as it is just not practical to kill all bacteria in the gut with these treatments.
Also – in another interesting avenue; Neuraminidases are used by viruses to multiply their infection rate in the host cells. In fact, Anti-viral drugs for Flu e.g. Tamiflu are neuraminidase inhibitors. This could potentially therefore be an explanation for CFS sufferers becoming susceptible to infection with and developing chronic forms of viruses such as EBV, CMV, Herpes Simplex etc. that other people clear (as described by many Physicians specialising in CFS, Eg Myhill, Cheney etc.). As higher circulating neuraminidases may benefit viruses and hamper the human immune system.
Finally, CFS is common, at over 1% of the population by most counts, so whatever causes it is more likely to be something that lots of people are exposed to like mild food borne pathogen exposure – rather than some rare pathogen that few people will be exposed to. So, it fits with the epidemiology.
The researchers prevented the condition developing with co-commutant supplementation with additional IAP – or a neuraminidase inhibitor. Both worked well, leading to the question of whether neuraminidase inhibitors – either natural forms (btw. I think turmeric may include them) – or pharmaceutical, could be useful in reversing CFS with gut symptoms in humans. Or IAP for that matter – there are some human trials of it in IBD already.
There is a summary here http://science.sciencemag.org/content/358/6370/eaao5610
Or the full study text here http://nizetlab.ucsd.edu/Publications/Gut-Inflammation.pdf
The study is in mice – so the usual provisos apply.
I wasn’t sure where was best to post this – but since Giardia is a gut pathogen i thought this would work – but feel free to move it.
I would be interested in others thoughts.
Intestinal Alkaline Phosphatase
- ” IAP is an endogenous protein expressed by the intestinal epithelium that is believed to play a vital role in maintaining gut homeostasis. Loss of IAP expression or function is associated with increased intestinal inflammation, dysbiosis, bacterial translocation and subsequently systemic inflammation. As these events are a cornerstone of the pathophysiology of many diseases relevant to surgeons,” [2016]
- “The present review analyzes the earlier literature on the dietary factors modulating IAP activity in light of these new findings. IAP regulates lipid absorption across the apical membrane of enterocytes, participates in the regulation of bicarbonate secretion and of duodenal surface pH, limits bacterial transepithelial passage, and finally controls bacterial endotoxin-induced inflammation by dephosphorylation, thus detoxifying intestinal lipopolysaccharide. Many dietary components, including fat, protein, and carbohydrate, modulate IAP expression or activity and may be combined to sustain a high level of IAP activity. In conclusion, IAP has a pivotal role in intestinal homeostasis and its activity could be increased through the diet. This is especially true in pathological situations (e.g., inflammatory bowel diseases) in which the involvement of commensal bacteria is suspected and when intestinal AP is too low to detoxify a sufficient amount of bacterial lipopolysaccharide.” [2010]
- “Intestinal alkaline phosphatase (IAP) plays an essential role in intestinal homeostasis and health through interactions with the resident microbiota, diet and the gut. IAP’s role in the intestine is to dephosphorylate toxic microbial ligands such as lipopolysaccharides, unmethylated cytosine-guanosine dinucleotides and flagellin as well as extracellular nucleotides such as uridine diphosphate. IAP’s ability to detoxify these ligands is essential in protecting the host from sepsis during acute inflammation and chronic inflammatory conditions such as inflammatory bowel disease. Also important in these complications is IAP’s ability to regulate the microbial ecosystem by forming a complex relationship between microbiota, diet and the intestinal mucosal surface. Evidence reveals that diet alters IAP expression and activity and this in turn can influence the gut microbiota and homeostasis. IAP’s ability to maintain a healthy gastrointestinal tract has accelerated research on its potential use as a therapeutic agent against a multitude of diseases. Exogenous IAP has been shown to have beneficial effects when administered during ulcerative colitis, coronary bypass surgery and sepsis. There are currently a handful of human clinical trials underway investigating the effects of exogenous IAP during sepsis, rheumatoid arthritis and heart surgery.” [2014]
SelfHacked.Com has an interesting post on this.
FYI, if you tried to…
FYI, if you tried to upload your uBiome in the last day and failed, please try again — there was a bug that has been fixed.
Comparing repeated uBiome results
There are some 15 readers who have done 2 or more uBiomes. I have added a new page to allow the people to see differences better.
After login at: http://microbiomeprescription.com/Biome/login a new choice will appear if you have done 2 or more uBiome:

Click this take you to a page to select which ones you wish to compare:

Selecting and clicking will take you to a page which will compare:
- Symptoms
- Function/Metabolites
- Bacteria
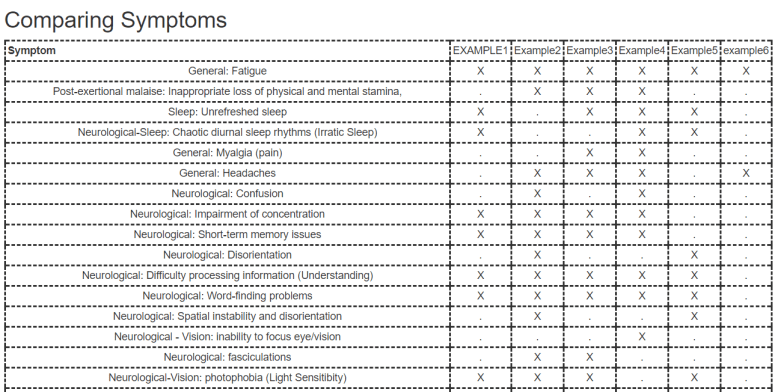
Comparing Function/Metabolism

Comparing Bacteria

Bottom Line
This tool will hopefully help with understanding how you have manipulated your microbiome by changes in diet, supplements, antibiotics and probiotics.
If you find interesting changes, feel free to post as a comment here.
