You may have a small heart
- “Cardiovascular symptoms are common in CFS patients. Cardiac dysfunction with low cardiac output due to small left ventricular chamber may contribute to the development of chronic fatigue as a constitutional factor in a considerable number of CFS patients.” [2009]
- “A considerable number of CFS patients have a small heart. Small heart syndrome may contribute to the development of CFS as a constitutional factor predisposing to fatigue, and may be included in the genesis of CFS.” [2008]
- Small heart syndrome in patients with chronic fatigue syndrome[2008]. “A considerable number of CFS patients have a small heart. Small heart syndrome may contribute to the development of CFS as a constitutional factor predisposing to fatigue, and may be included in the genesis of CFS.”
- “A small size of LV with low cardiac output was noted in OI, and its degree was more pronounced in CFS with Orthostatic intolerance CFSOI(+). A small heart appears to be related to the genesis of OI and CFS via both cerebral and systemic hypoperfusion. CFSOI(+) seems to constitute a well-defined and predominant subgroup of CFS.”[2011]
- “”Small heart” on the chest X-ray photograph was prevalently noted in CFS patients. Echocardiographic examination revealed that CFS patients with “small heart” had an actually small LV chamber and poor cardiac performance.” [2009]
- From “Small Heart as a Constitutive Factor Predisposing to Chronic Fatigue Syndrome [2012]”(Full Text)
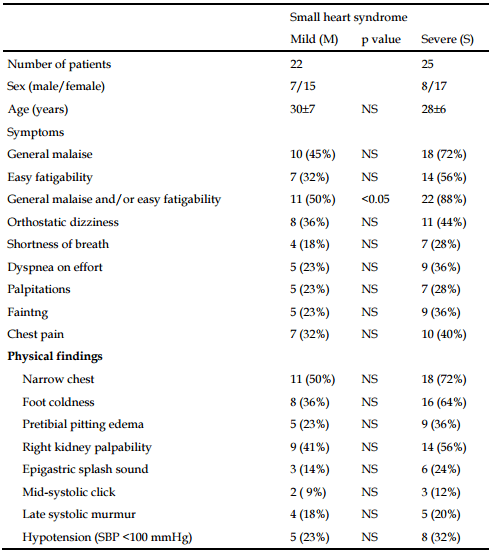

Rapid Heart Beat (Tachycardia) and POTS
A consequence of a small heart may be a tendency to increase blood flow to the body by beating faster. The small heart may also be connected to the DNA SNPs associated with CFS.
- “The heart rate dynamic response during the head-up tilt test differs between patients with CFS and healthy controls, supporting the increased prevalence of postural orthostatic tachycardia syndrome.” [2014]
- Painful temporomandibular disorders are common in patients with postural orthostatic tachycardia syndrome and impact significantly upon quality of life[2015].
- Postural tachycardia syndrome is associated with significant symptoms and functional impairment predominantly affecting young women: a UK perspective [2014]. “Patients with PoTS are predominantly women, young, well educated and have significant and debilitating symptoms that impact significantly on quality of life.”
- Postural neurocognitive and neuronal activated cerebral blood flow deficits in young chronic fatigue syndrome patients with postural tachycardia syndrome [2012].
- ncreasing orthostatic stress impairs neurocognitive functioning in chronic fatigue syndrome with postural tachycardia syndrome [2012].
- “A substantial number of CFS patients have orthostatic intolerance in the form of orthostatic hypocapnia[a state of reduced carbon dioxide in the blood].”[2007]
- Patterns of hypocapnia on tilt in patients with fibromyalgia, chronic fatigue syndrome, nonspecific dizziness, and neurally mediated syncope [2006]. “Hypocapnia was diagnosed on tilt test in 9% to 27% of patients with fibromyalgia, CFS, dizziness, and NMS versus 0% to 2% of control subjects.”
- “Postural orthostatic tachycardia syndrome (POTS) is a heterogeneous disorder of the autonomic nervous system in which a change from the supine position to an upright position causes an abnormally large increase in heart rate or tachycardia (30 bpm within 10 min of standing or head-up tilt). This response is accompanied by a decrease in blood flow to the brain and hence a spectrum of symptoms associated with cerebral hypoperfusion. Many of these POTS-related symptoms are also observed in chronic anxiety and panic disorders, and therefore POTS is frequently under- and misdiagnosed.” [2016]
Blood Characteristics
For a good discussion on blood differences seen in CFS, see Les Simpson article in the Journal of IiME, Vol. 2 Issue , p. 24ff.
- “Echocardiographic measures indic ated that the severe CFS participants had 10.2% lower cardiac volume (i.e. stroke index and end-diastolic volume) and 25.1% lower contractility (velocity of circumferential shortening corrected by heart rate) than the control groups.” [2009]
- ” In conclusion, individuals with CFS have a significantly lower peak oxygen consumption and an insignificant trend toward lower blood volume compared with controls. These variables were highly related in both subject groups, indicating that blood volume is a strong physiological correlate of peak oxygen consumption in patients with CFS.” [2002]
- “Circulating Blood Volume in Chronic Fatigue Syndrome [1998]” ” Of the 19 patients reported here, abnormalities in blood volume were very common. The most common, found in 16 of 19 patients, was a reduction in red blood cell mass. Eleven subjects had low plasma volumes, and total circulating blood volume was subnormal in 12 of 19 subjects. In some individuals this abnormality was strikingly severe… red Blood Cell mass …46% of the expected normal, and a total blood volume ..which represents 49.7% of the expected normal value”
-
- Less and slower variability of blood pressure (2012) (2011)
- Lower total blood volume(8%(2009) – 9% (2002) – 15%(2009) less), plasma volume (13% (2009)) and red blood cell volume (19%) (2009)(2000)(2007)(1998).
- 35% lower peak oxygen consumption(2002)
- Significant decrease in red cell distribution width (2007)
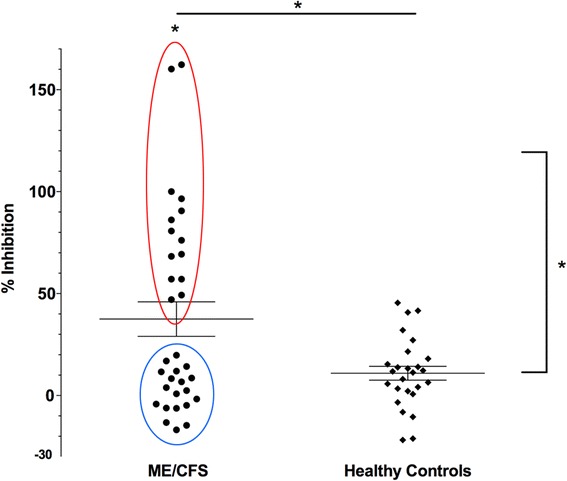
- Higher percentages of misshaped red blood cells[13] [14] (2001) (other)
- Impaired capillary blood flow.
- Changed red cell shape populations
- High values for flat blood-cells
- Percentage of deformed cells reduced with B12 injections within 24 hrs in responders (P. 245 Englebienne, P. (2002). Chronic Fatigue Syndrome: A Biological Approach.)
And now add thick blood!
- Chronic fatigue syndrome and/or fibromyalgia as a variation of antiphospholipid antibody syndrome: an explanatory model and approach to laboratory diagnosis [ 1999]. “CFS and/or FM patients who have a hereditary deficiency for thrombophilia or hypofibrinolysis may be unable to control thrombin generation properly. We have found that three out of four CFS and/or FM patients have a genetic deficiency (unpublished data). Certain pathogens induce the immune system generation of APL antibodie s and can be a triggering mechanism for APS. Once antibodies are formed, protective proteins are dislodged from endothelial cells, exposing PS. Coagulation proteins bind on exposed PS surfaces, generating thrombin on the EC surface. Excess thrombin converts fibrinogen to SFM, which may be deposited on the EC surface and/or circulate in the plasma. Fibrin deposition leads to decreased oxygen, nutrient and cellular passage to tissues around the microcirculation. This hypercoagulable state may cause localized pathology in many tissues, yielding the systemic compromises and symptoms characteristic of the CFS-FM complex. Since this hypercoagulable state does not necessarily result in a thrombosis, but rather in fibrin deposition, we suggest that an alternative name for this Antiphospholipid antibody process would be immune system activation of coagulation (ISAC) instead of antibody-mediated thrombosis [18]. Once this hypercoagulable state is detected, appropriate anticoagulant therapies may be given to relieve patient symptoms
- “Most symptoms of Gulf War Illness (GWI) are similar to Chronic Fatigue Syndrome (CFS) and/or Fibromyalgia (FM). We investigated whether these symptoms are associated with an activated coagulation system as has been reported in some cases of CFS/FM. The coagulation assays include activation markers of the cascade, platelet activation and hereditary risk factors. Our findings show activation of the coagulation system in GWI. This evidence of a hypercoagulable state suggests that symptoms may be due to poor blood flow and, therefore, a basis for the potential utility of anticoagulant therapy.” [2000]
Bottom Line
Recipe for CFS: Start with a small heart, then decrease it’s efficiency by having thicker blood and less blood, add in alterations of metabolites (amino acids and other chemicals) due to microbiome shifts — adding more chaos. Filter thru your DNA Snps to produce your own special collection of symptoms!
In terms of treatment:
- Fix metabolites
- Reduce coagulation / thick blood
Heart Problems?
- Why myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) may kill you: disorders in the inflammatory and oxidative and nitrosative stress (IO&NS) pathways may explain cardiovascular disorders in ME/CFS [2009].
- Coenzyme Q10 deficiency in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is related to fatigue, autonomic and neurocognitive symptoms and is another risk factor explaining the early mortality in ME/CFS due to cardiovascular disorder [2009].
- Lower plasma Coenzyme Q10 in depression: a marker for treatment resistance and chronic fatigue in depression and a risk factor to cardiovascular disorder in that illness [2009].
- Causes of death among patients with chronic fatigue syndrome [2006]. “The three most prevalent causes of death were heart failure, suicide, and cancer, which accounted for 59.6% of all deaths.”