In my last few posts I looked at illnesses in the autoimmune spectrum that have started to have their microbiome studied. For each of them I used DataPunk.net to filter out good and bad things for correcting the shifts done. I then spot checked some of the things against the literature and discovered that the predictions of what could happen — frequently was found to help. The oddest one was that using diet-pop (versus regular pop) reduced the risk of Rheumatoid Arthritis was confirmed and matched the prediction that sugar substitutes helped correct/prevent this specific microbiome shift.
At present I do believe that CFS is likely a mixture of different microbiome shift patterns. This comes from two things:
- the definition requires X of Y symptoms and symptoms are related to the microbiome shifts
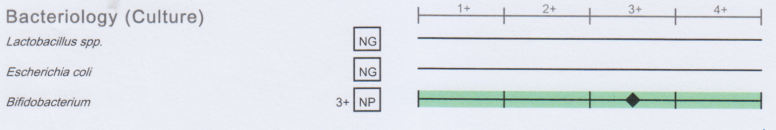
- going thru many uBiome results — I see the commonality of low or no lactobacillus, bifidobacterium and e.coli (originally reported in 1998) but with all of the other bacteria genus being all over the place.
We can shift microbiome patterns by diet. The problem is that the diet should match the microbiome shift desired. This means that any specific diet as a general recommendation to CFS patients becomes suspect without a detail examination of the microbiome first. Eating Navy beans and barley oatmeal for breakfast will help some and hurt others.
A reader asked explicitly about the paleo-ketogenic diet, so I will start by summarizing what research reports.
Paleo-ketogenic diet Research
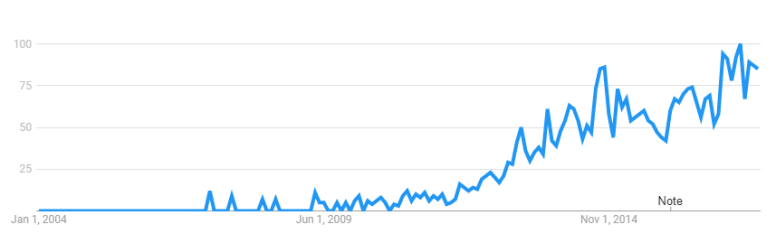
Google Trends shows us that it started around 2009 and has been growing in interest.
Going to PubMed – Nothing for this search term. On Google Scholar, we had 681 hits (often blog posts). I used the following from the Ancestral Health Society’s “Annual symposium of the German Society for Paleo Nutrition”
- http://jevohealth.com/journal/vol1/iss1/5/ (2013)
- http://jevohealth.com/journal/vol1/iss1/6/ (2014)
- http://jevohealth.com/journal/vol1/iss1/8/ (2015)
With the following being asserted:
“Adjusting to the ketogenic Paleo diet led to subjectively noticeable health benefits:
• Significant weight loss of 70 kg (154 pounds) within 15 months
• Enhanced metabolic fat burning and gains of lean body mass
• Improved cognitive performance
• Stronger immune system
• Mental balance and general well-being” 2013, p7
Bottom line, I was unable to find any clinical studies.
What is the diet and how does it impact the microbiome?
- The ketogenic diet is a high-fat, adequate-protein, low-carbohydrate diet [Wikipedia]
- The Paleolithic diet (also called the paleo diet, caveman diet or stone-age diet[1]) is a modern fad diet requiring the sole or predominant consumption of foods presumed to have been the only foods either available or consumed by humans during the Paleolithic era [wikipedia]
- “the diet typically includes vegetables, fruits, nuts, roots, and meat while excluding foods such as dairy products, grains, sugar, legumes, processed oils, salt, and alcohol or coffee.[1] The diet is based on avoiding not just modern processed foods, but rather the foods that humans began eating after the Neolithic Revolution when humans transitioned from hunter-gatherer lifestyles to settled agriculture.”
The wikipedia definitions actually provide us with sufficient information to infer what expected shifts in the microbiome may be due to our friend, DataPunk.Net.
Ketogenic Diet – simply BAD for CFS
A quick trip to DataPunk.net revealed that it inhibits Lactobacillus and double inhibits Bifidobacterium. Forget all of the other bacteria genus — it’s wrong for the basics!!!!
Addenum:
UPDATE: I just checked the old CfsFMExperimental Group on Yahoo and the use of ketogenic diet was a topic on that group back in 2000-2005. It never went anywhere — although people tried it.
Paleolithic diet
- High fruit intake: Enhances Alistipes
- Plant Rich Diet: Inhibits Alistipes
- Almonds/ almond skins: Enhances Bifidobacterium, Lactobacillus
- Walnuts:
- Enhances: Bacilli Coprococcus Firmicutes Lachnospiraceae Lactobacillus reuteri Oscillospira Peptococcaceae Roseburia Ruminococcaceae Streptophyta Turicibacter
- Inhibits:Alphaproteobacteria Anaeroplasmatales Anaerotruncus Bacteroides Bacteroidetes Blautia Carnobacteriaceae Dehalobacter Erysipelotrichia Gammaproteobacteria Proteobacteria Tenericutes
- High animal protein diet
- Sesame seed: Enhances Bifidobacterium Enterococcus Lactobacillus
Compared to the ketogenic diet, the Paleolithic diet is a winner. Lean animal meat is in keeping with paleolithic life (all animals were wild and hunted by men and other creatures).
Bottom Line
I prefer a diet custom tuned to the microbiome shifts — but if you want a ‘stock diet’ I would prefer you to consider a paleolithic diet (although it is vaguely defined).