A reader messaged me the following summary(in full). This interests me and as a result, this is a long post as I explore her before and after microbiomes.
Case History: 4hr Biochemically Mediated Spontaneous Remission of Environmental Sensitivity CFS Symptoms
I experienced a total remission of the brain fog/environmental sensitivity and sensory sensations that accompanies severe CFS for ~4 hours on April 23rd, 2019 and then slowly slipped back in.
It was a biochemical/microbiome spontaneous remission which occurred following treatment with probiotics (VSL#3) and BCAAs as part of a liquid meal drink (ghee, MCT oil) (thus affecting gut signaling of tryptophan) after I used antibiotics (Amoxicillin And Clavulanate) to treat an infected dental abscess.
It was unintentional as I was just trying to repopulate my microbiome after 3 day antibiotic use and was trying to get high quality nutrients in liquid form as the wound in my mouth made it difficult to eat. The ‘fog’ lifted within about 10 minutes. It was shocking and scary and wonderful. I realised just how sick i was/am and this was a massive shock. I could feel the world again, wanted to feel the world again, and at the same time was overwhelmed with just how different i felt from my usual sick state. It felt like a miracle – and it was, a biochemically triggered miracle.
I was in this state for about 2 hours and then slowly started slipping back into the fog and illness. I took more VSL#3 and a tryp-lowering drink (mix of honey or agave, coconut milk, mct oil or ghee, vegan protein powder, and turmeric or matcha) but i continued to sink back into the CFS state.
The next day i took the same mix of substances but no phase shift occurred. I knew what happened was amazing but also dangerous and that i should try and re-stabilize to baseline. Ironically, I had watched Robert Phair’s talk that morning on springing the metabolic trap through the tryptophan-kynurenine pathway.
I have been following Ron Davis’ group/Robert Phair/Robert Naviaux the Open Medicine Foundation’s research and understood the basics of the metabolic trap hypothesis but was not trying to ‘spring the trap’, i knew that this could actually very dangerous and got scared and contacted Dr Phair-he confirmed the likely timescale of the event and was not worried about trying to re-create it.
This experience provided a case history w/ clinical evidence for the role of the tryp-kyn pathway in springing the metabolic trap. It is hard to assess the severity of one’s own CFS but i would guess that i am in the bottom 25% in terms of severity but am only 95% bedridden (I get out for specialist MD appts but that is about it). So not the worst of the severe cfs patients but maybe on the cusp. I am obviously excited about this event and think it will be of interest to cfs researchers but am also quite scared that i could accidentally make myself worse.
I am very much hoping that my case could be of interest and use to cfs research and others. I have had my genome sequenced (WGS thru Veritas – I have ID02 mutations) and extensive biochemical workups have shown high 5-HIAA and serotonin as well as high levels of VLCFAs suggestive of mitochondrial and peroxisomal dysfunction.
I believe the CFS component was triggered initially in 2002 after getting dengue fever. in 2008 and 2013 traumatic life events occurred which led to steadily progressive fatigue and pain. By 2016 i was on 100% disability leave and i was dx’d with hEDS in 2017. I have tried to spring the trap since without success.
I had a ubiome sample pack the day the remission occured and took a stool sample the next morning.
- Apr 11/2019 Before
- Apr 24/2019 Remission
Time Lines
- Prior to 11th, herbals: Cleavers, Echinacea, Goldseal, Gotu Kola, Lemon Balm, Marigold, Pau D’arco, Thuja, Usnea Lichen, Bearberry, Buchu, Corn Silk Stigma, Couch Grass, Goldenrod, Goldenseal, Horsetail, Marshmallow, Oregon Grape
- 19th-22nd Amoxicillin And Clavulanate)
- 22-24th VSL#3
Tryp-Kyn Pathway
This is the model/hypothesis that the reader is attempting to apply to CFS/ME. There is a good overview article (2017).
The microbiome model fits around this model (and other models), because the amount of tryptophan, etc, is recognized to be influenced by the microbiome. This is well stated in the 2018 article below (and others)
The gut microbiota is a crucial actor in human physiology. Many of these effects are mediated by metabolites that are either produced by the microbes or derived from the transformation of environmental or host molecules. Among the array of metabolites at the interface between these microorganisms and the host is the essential aromatic amino acid tryptophan (Trp). In the gut, the three major Trp metabolism pathways leading to serotonin (5-hydroxytryptamine), kynurenine (Kyn), and indole derivatives are under the direct or indirect control of the microbiota. In this review, we gather the most recent advances concerning the central role of Trp metabolism in microbiota-host crosstalk in health and disease. Deciphering the complex equilibrium between these pathways will facilitate a better understanding of the pathogenesis of human diseases and open therapeutic opportunities.
Gut Microbiota Regulation of Tryptophan Metabolism in Health and Disease. [2018]
My take is to keep the view simpler, instead of trying to determine the complex equilibrium — just identify which bacteria combinations are causing the equilibrium to be off. How do we do this? By identifying combinations strongly statistically associated with symptoms! (see this post) Once the bacteria are identified, then proceed to alter their population with modifiers (supplements, diets, prescription drugs, probiotics, flavonoids, etc). With this view, we can take action today. The alternative is to wait until they have deciphered it, then wait again while they attempt to determine and test strategies to adjust the equilibrium…. actions may be years, it not decades off.
Initial Overview
There were no lab measurements of items in the Tryp-Kyn pathway before and at this remission. So using that model would be speculation. There are microbiome measurements and thus we have some concrete lab results to look for an explanation of this short term remission. Hopefully, we will also get microbiome results after the relapse to see more of the pattern. Going to the sample pages and then clicking compare samples takes us to a bunch of useful pages.
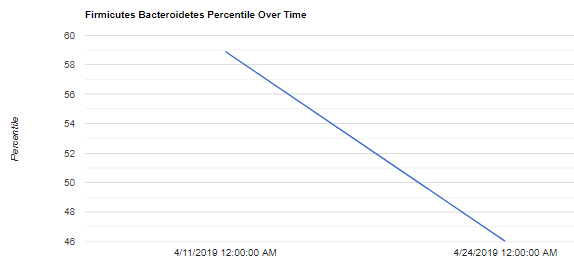
At a high level we see a significant change of Firmicutes to Bacteroidetes ratio
What else has changed at a high level? Walking thru the Class level we see the following major changes:
- Alphaproteobacteria went from down to 1/36 (from high to very low percentile)
- Epsilonproteobacteria went up 30x (from median to high percentile)
- Gammaproteobacteria went from nothing to very high percentile
- Opitutae went from high percentile to none
- Tissierellia went up 30x (from high to very high)
- Verrucomicrobiae moved from none to the median levels
There was a lot of changes at each of the different taxonomy ranks. Not unexpected as a result of Amoxicillin And Clavulanate (these are linked to details of what they changed). A lot of changes makes interpretation complex.
Manual Data Analysis
Since the antibiotic impact data is at species, genus and family only, I decided is to see how the predicted shifts (from pub med studies) match up with actual shifts in two ways:
- Before + after: with Amoxicillin,Clavulanate and VSL#3
- Before + after: with Amoxicillin,Clavulanate
Most of the data on the impact on bacteria by antibiotics came from one study. This study has a known weakness of excluding interplay between bacteria. A second factor to consider is that there can be a time delay for shifts to appear. With my own flare, I saw some immediate drops, other bacteria did a slow decline and still others did not start to decline until 2-3 weeks later. Bacteria behavior and population will change only when the circulating level of metabolites changes. It is a long supply chain. Applying tariffs on lumber leaving on ships to India does not immediately cause the price of lumber in India to jump. As ships arrive and existing supplies are consumed, we will see the price increase.
The remission microbiome sample was done on day 5 after starting antibiotics, and 2 days after stopping. We need to check the half life for these antibiotics to see if they are still active. We find that they should be almost zero left when the remission started..
- Amoxicillin: 60% is excreted after 6-8 hrs [src]
- Clavulanate: half life is 1.3 hours
IMPORTANT NOTE: The antibiotics may have started a cascade (destabilizing the microbiome) – from observations on my own microbiome, these can continue on for weeks until a new stable state is realized. Think of supply chains!
Amoxicillin,Clavulanate and VSL#3
Reminder: the numbers are created by summing up the number of studies reporting increases and decreases. They do not represent the magnitude of the changes (which is rarely given). A number like 0.1 suggests that one of the above increase it in some studies, another one may decreases it (or a different study reported different results). Yes, it s@cks, but that is the state of the medical literature. We do the best with what we have.
Family Rank
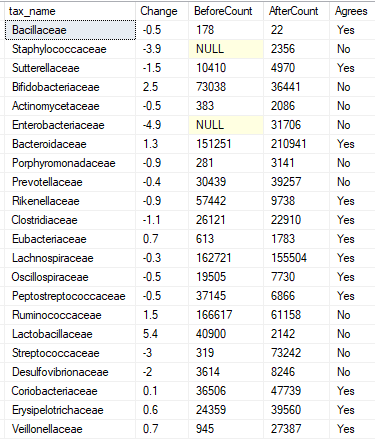
Genus Rank
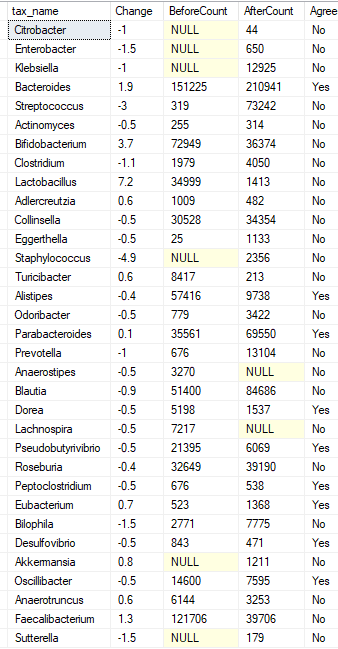
Species Rank
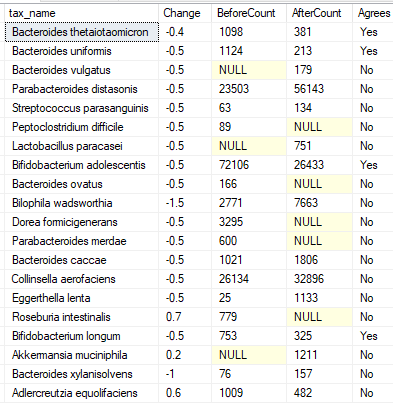
Amoxicillin,Clavulanate
Family
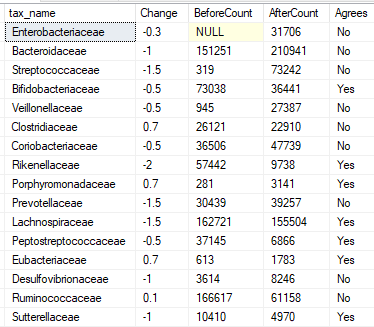
Genus
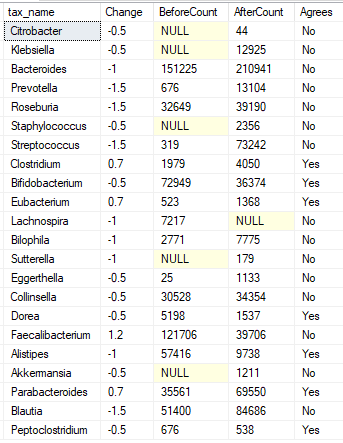
Species
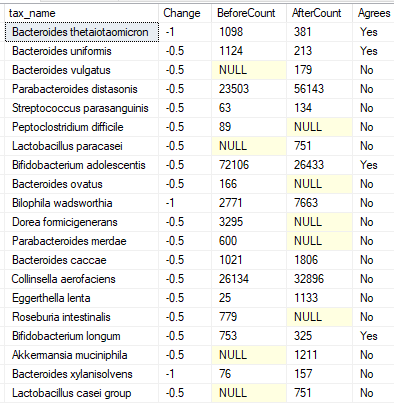
Review of the Above
One item that stood out for me was the increase of Veillonellaceae family, this is the family that is very high in world class athletes (see this post). It increased by 27x times (and moved into the top 75%ile). This bacteria consumes lactate [ The technical difference between lactate and lactic acid is chemical. Lactate is lactic acid, missing one proton]. Lactic acidosis is often reported for ME/CFS and that is strongly associated with brain fog).”This bacteria turn lactate into propionate, a short-chain fatty acid that’s a source of energy and can also be an anti-inflammatory ”
This change alone could account for the temporal relapse. A subsequent microbiome is in the works — if Veillonellaceae has dropped down to prior levels — we have a viable model for why the remission happen. The logical thing would be shift to a Veillonella friendly diet and supplements. Note that the VSL#3 inclusion shifted the predicted impact from negative to positive.
Keeping with the probiotic approach, we find the following reported to increase Veillonella (other probiotics decreases it):
- bifidobacterium animalis subsp. lactis (probiotics)
- bifidobacterium longum (probiotics)
- lactobacillus acidophilus (probiotics)
Further Analysis
Looking at the summaries, the person appear to have gotten worst. Remember these are rough predictors.

These samples were taken some 13 days apart. At a high level we see things suggesting worst, not better. Hmmm…
- 4/11/2019 – (Before) had 11 predicted symptoms
- 4/24/2019 – (Remission) had 22 predicted symptoms
The shared items are below.
- Autonomic Manifestations: Cortisol disorders or irregularity
- Immune Manifestations: Abdominal Pain
- Neurological: Disorientation
- Neurological: Neuropathy
- Neurological: Slowed speech
- Onset: less than 16 years since onset
- Post-exertional malaise: Post-exertional malaise
There is an interpretation challenge because of the short duration of the remission. It is unclear if we are actually measuring the remission microbiome or post-remission microbiome.
De-Lactic Acidosis happening?
The Veillonellales shift interests me. We do not see any actual Veillona genus, but do see other members. The Dialister propionicifaciens interests me because the name implies it produces very significant propionate (likely from lactic acid as does its cousin, Veillona). I was unable to find more definitive information about it.
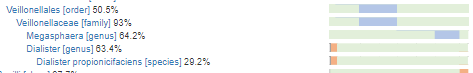
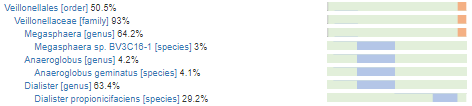
Checking out this family, we see that many CFS associated symptoms occur with the lowest levels. When we had our short remission — the numbers were at the top.

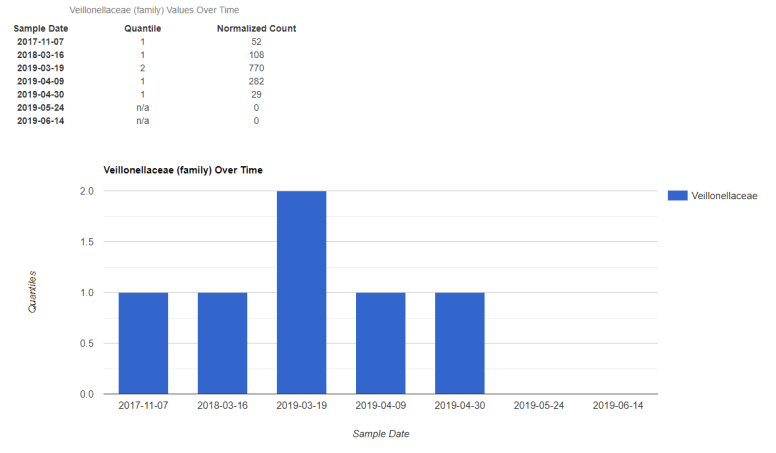
Running with the Veillonella vector
I am almost totally recovered from a flare — except for one very frustrating issue: I have 10-20 minute of physical activity limits (walking ~ 4 blocks, driving ~ 20 min). No brain fog. No apparent GI issues etc. This was not there at the start of the flare and really became pronounced around May 14th. I decided to pull up my own history and behold! My levels hit zero and stayed at zero at that point. Low levels have also been reported in a 2018 PubMed study.
Bottom Line
I spent a day trying to tease other information out. There are some wild swings, From bottom outlier to top outliers!) for items like Streptococcus I reach the point that I was trying to force the analysis too much.

This post demonstrate that antibiotics can really shake up the gut. As shown above, some bacteria taxonomy may vanish and other may appear.
My impression is that in the chaos caused by the antibioitics, VSL#3 supported one faction in the massive chaos of bacteria sufficiently for a short lived ‘functional government’ to come into existence before the neighboring chaos overwhelmed it.
The key thing is to remember that the microbiome function as a supply chain. A long supply chain. Impacting one species alters the environment which then respond, causing more changes that may take weeks to stabilize.
This appears to be confirmed by the dramatic change in different end products (experimental page) seen between the two samples:
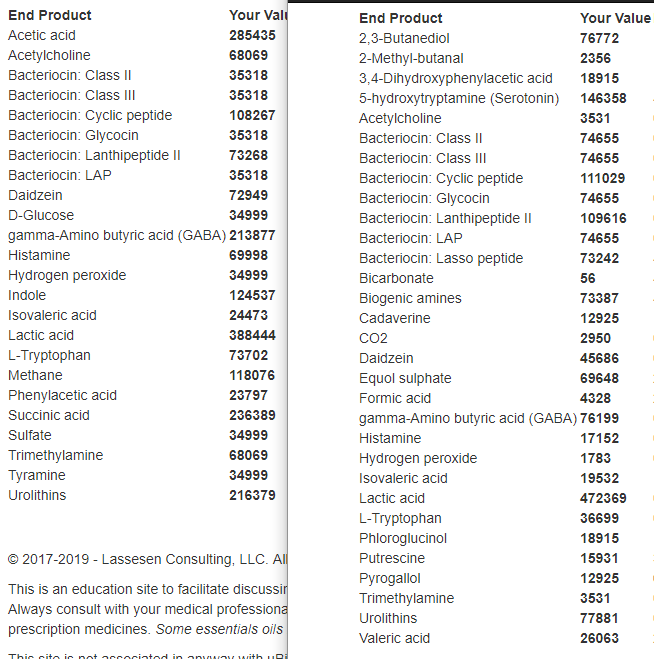
I speculate that this short remission happened because lactic-acid production dropped (granting relief to lactic acidosis) with a sudden increase in lactic acid consumers that produce propionate giving energy.
This study suggests a probable link between intestinal colonization of Gram positive facultative anaerobic D-lactic acid bacteria and symptom expressions in a subgroup of patients with CFS. Given the fact that this might explain not only neurocognitive dysfunction in CFS patients but also mitochondrial dysfunction, these findings may have important clinical implications.
Increased D-Lactic Acid Intestinal Bacteria in Patients with Chronic Fatigue Syndrome [2009] “
I look forward to add the relapse microbiome into the analysis.
