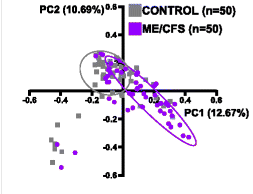
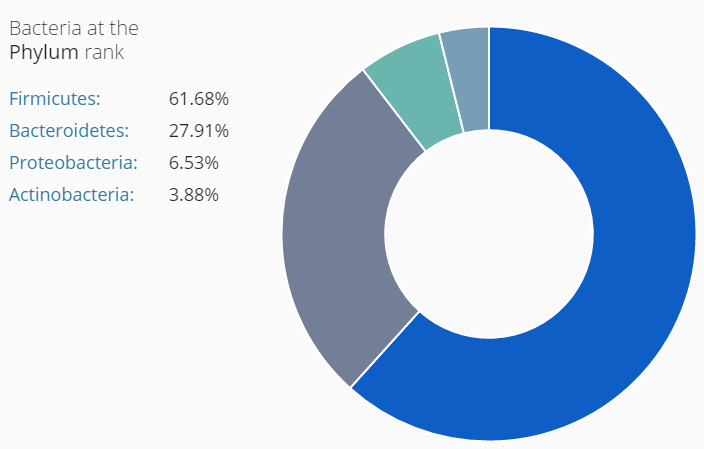
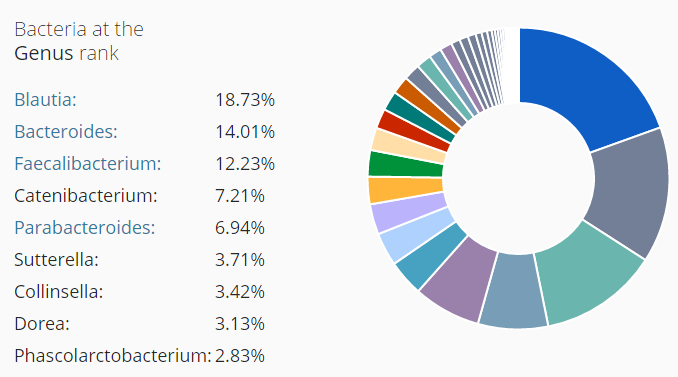
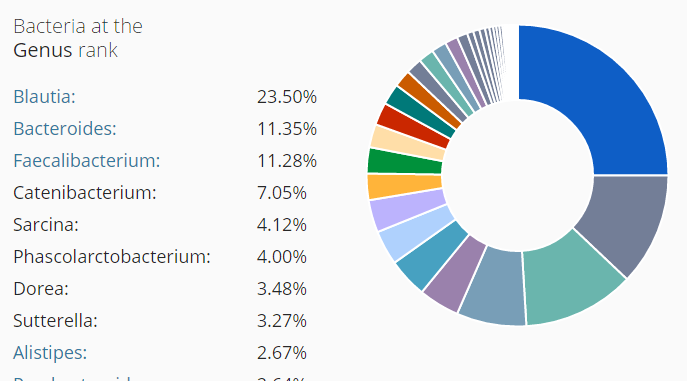
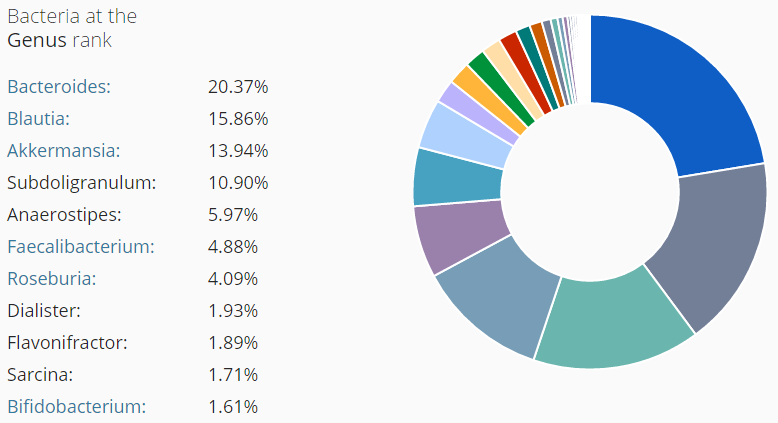
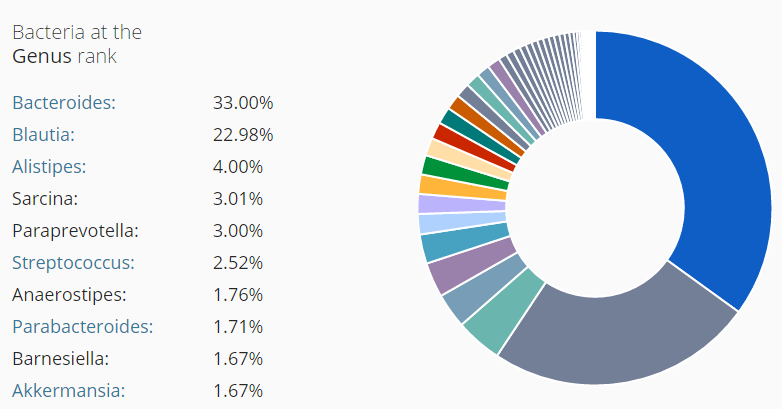
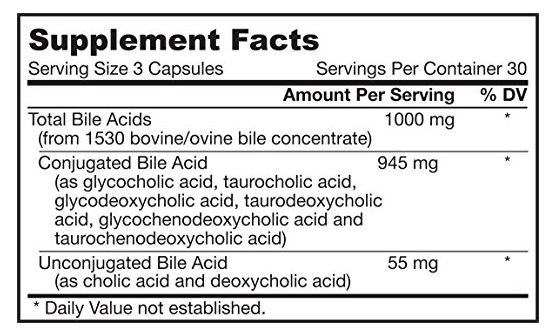
I did a post earlier on a CFS patient whose labs reported high level (a subset, possible very small) and what could be done to reduce a specific strain. Ian Lipkin study found (on average) low levels for:
- Faecalibacterium prausnitzii
- Faecalibacterium cf
Some microbiome results have high faecalibacterium, to reduce it, see this post https://atomic-temporary-42474220.wpcomstaging.com/2017/10/11/reducing-faecalibacterium-genus/ (that page will be updated regularly when new studies are published).
For updated information see Microbiome Prescription
This page is no longer being updated.
UPDATED INFO:
- Faecalibacterium OverGrowth Undergrowth
- Faecalibacterium prausnitzii Overgrowth Undergrowth
I will look at the faecalibacterium [Index to all posts on Study]. It’s relationship is shown below

Faecalibacterium (formerly Fusobacterium) prausnitzii (1995) is one of the three most abundant species detected in human feces by anaerobic cultivation (1995) and consists of many stains [2012] as shown below. As with all families, there can be some black sheep: Faecalibacterium prausnitzii subspecies-level dysbiosis in the human gut microbiome underlying atopic dermatitis [2016].

Items to Increase (take)
- Prescription
- “Rifaximin appeared to influence mainly potentially detrimental bacteria, such as Clostridium, but increasing the presence of some species, such as Faecalibacterium prausnitzii.” [2015] [2011]
- “α-glucosidase inhibitor acarbose …48 OTUs increased by 12.8-fold, including Lactobacillaceae (8 of 9 belonging to Lactobacillus), Ruminococcaceae (6 of 11 belonging to Faecalibacterium)” [2017]
- Probiotics
- Probiotic appears available for veterinarians [2017] [2012] [2015]
- “Bacillus coagulans GBI-30, 6086 (BC30); GanedenBC(30)] …increased populations of Faecalibacterium prausnitzii” [2015] [2014]- note: this increases histamine also
- Enhanced butyrate formation by cross-feeding between Faecalibacterium prausnitzii and Bifidobacterium adolescentis.
- “Bifidobacterium adolescentis L2-32, which produces acetate during fermentation, and Faecalibacterium prausnitzii A2-165, which consumes acetate and produces butyrate.” [2014] so acetate producing probiotics are likely good.
- Enterococcus durans EP1 a Promising Anti-inflammatory Probiotic Able to Stimulate sIgA and to Increase Faecalibacterium prausnitzii Abundance [2017]. Note this was the only one of 15 Enterococcus faecium studied that had this effect. — Very strain specific. Researchers are in France.
- “Bifidobacterium pseudocatenulatum in fecal cultures from some individuals accounted for the prevalence of Desulfovibrio and Faecalibacterium prausnitzii,” [2008]
- oligosaccharides /fibre or similar
- “The ranking order of butyrate production rates was amylopectin > oat xylan > shredded wheat > inulin > pectin (continuous cultures), and inulin > amylopectin > oat xylan > shredded wheat > pectin (batch cultures). The contribution of external acetate to butyrate formation in these experiments ranged from 56 (pectin) to 90 % (xylan) in continuous cultures” [2004] – Faecalibacterium consumes acetate and produces butyrate.
- “Relative abundance of lactose-fermenting Bifidobacterium, Faecalibacterium, and Lactobacillus were significantly increased in response to short-chain galactooligosaccharide.” [2017]
- “soybean oligosaccharides..increased the diversity of intestinal microflora and elevated (P < .05) the numbers of some presumably beneficial intestinal bacteria [2014]
- “inulin stimulated colonization by Faecalibacterium prausnitzii, which has anti-inflammatory effects,”[2009] [2013] 10% increase [2009]
- “Kestose[a fructooligosaccharide] efficiently stimulates the growth of this bacterium in the intestine,” [2016]
- “numbers of lactobacilli, bifidobacteria (P<0.001) and Faecalibacterium prausnitzii (P<0.05) were higher in the low-fat/high-fiber pigs than in high-fat/low-fiber pigs,” [2016] [2016]
- “Associated growth of levan-degrading (e.g. Bacteroides) and butyric acid-producing (e.g. Faecalibacterium) taxa was observed in levan-supplemented media.” [2015]
- “Potato fibres [PF]…increases in the proportion of Faecalibacterium (not Lactobacillus/Bifidobacterium, as confirmed by qPCR analysis) with increasing dietary PF concentrations suggest that PF is a possible prebiotic fibre.” [2015]
- “Resistant maltodextrin (RM)..was associated with statistically significant increases (P < 0.001) in various operational taxonomic units matching closest to ruminococcus, eubacterium, lachnospiraceae, bacteroides, holdemania, and faecalibacterium, implicating RM in their growth in the gut.” [2014]
- “resistant starch… increased the relative abundance of several butyrate-producing microbial groups, including the butyrate producers Faecalibacterium prausnitzii and Megasphaera elsdeni” [2013] [2013]
- “arabinogalactan… was associated with a statistically significant increase in the concentration of total bacteria, Bacteroidetes, Faecalibacterium prausnitzii” [2013]
- “Isoflavones[beans] alone stimulated dominant microorganisms of the Clostridium coccoides-Eubacterium rectale cluster, Lactobacillus-Enterococcus group, Faecalibacterium prausnitzii subgroup, and Bifidobacterium genus.” [2005]
- “sodium butyrate increased the relative abundance of Bacteroides and Faecalibacterium” [2016] – Faecalibacterium produces butyrate, so there appears to be a feed back cycle here.
- Diet
- “after a specific carbohydrate diet, there was an increased representation of Faecalibacterium prausnitzii, an anti-inflammatory commensal.”[2016]
- “Subjects with higher adherence to a Mediterranean dietary pattern presented…higher levels of Clostridium cluster XVIa and Faecalibacterium prausnitzii.” [2017] [2016]
- “Vegetarian diet was associated with higher ratio (% of group-specific DNA in relation to all bacterial DNA) of Bacteroides-Prevotella, Bacteroides thetaiotaomicron, Clostridium clostridioforme and Faecalibacterium prausnitzii, but with lower ratio (%) of Clostridium cluster XIVa…Up to 4 % of variance in microbial community analyzed .. could be explained by the vegetarian type of diet.” [2014]
- “high high carbohydrate/low glycemic index and high saturated fat diet increased Faecalibacterium prausnitzii” [2013]
- Increased gut microbiota diversity and abundance of Faecalibacterium prausnitzii and Akkermansia after fasting: a pilot study [2015].
- Foods
- “The relative abundance of Akkermansia and Faecalibacterium, genera associated with healthy gut mucosa and anti-inflammation, was found to increase in response to lingonberry intake.” [2016]
- “cocoa husks …increased the Bacteroides-Prevotella group and Faecalibacterium prausnitzii,” [2016]
- “Broad beans (Vicia faba) and lupin seeds (Lupinus albus) are legumes rich in a wide range of compound … increased significantly (P < 0.05) were Bifidobacterium spp., Lactobacillus-Enterococcus, Atopobium, Bacteroides-Pretovella, Clostridium coccoides-Eubacterium rectale, Faecalibacterium prausnitzii and Roseburia intestinalis.” [2015]
- “ red wine polyphenols significantly increased the number of fecal bifidobacteria and Lactobacillus (intestinal barrier protectors) and butyrate-producing bacteria (Faecalibacterium prausnitzii and Roseburia) at the expense of less desirable groups of bacteria such as LPS producers (Escherichia coli and Enterobacter cloacae).” [2016] i.e. RED Grape Seed Extract
- “dihydroflavonols were directly associated with Faecalibacterium … red wine was the best contributor to Faecalibacterium variation.” [2015]
- “Most F. prausnitzii strains tested grew well under anaerobic conditions on apple pectin…..Many F. prausnitzii isolates were able to utilize uronic acids for growth …Most strains grew on N-acetylglucosamine” [2012]
- “Dietary capsaicin [cayenne peppers] increased the Firmicutes/Bacteroidetes ratio and Faecalibacterium abundance,” [2016]
- “Curcuma longa[turmeric] and Scutellaria baicalensis [Chinese Skullcap] used as feed additives…S. Enteritidis, Faecalibacterium, and Lactobacillus, both bacterial genera with known positive effects on gut health were positively selected” [2015]
- “was more abundant in the raffinose[beans, etc] diet and the chickpea diet” [2010]
- “MSG promoted the colonization of Faecalibacterium prausnitzii” [2015]
- “several experiments involving downshifts to pH 5.5 resulted in Faecalibacterium prausnitzii replacing Bacteroides spp. as the dominant sequences observed.” [2016]
- “correlation to vacuum cleaning frequency, with an increase in Faecalibacterium prausnitzii for mothers” [2015]
Items with no effect
Items that decrease (avoid)
- :Metronidazole… known to decrease anaerobes such as Faecalibacterium” [2016]
- “This case study investigated changes of gut microbiota with an omega-3 rich diet. Fecal samples were collected from a 45-year-old male who consumed 600 mg of omega-3 daily for 14 days. After the intervention, species diversity was decreased, but several butyrate-producing bacteria increased. There was an important decrease in Faecalibacterium prausnitzii and Akkermansia spp. Gut microbiota changes were reverted after the 14-day washout.” [2016]
- “proton pump inhibitor…was accompanied by a lower abundance of Faecalibacterium spp.” [2012]
- “antibiotics [which ones not specified] most notably a decrease in relative Faecalibacterium spp. numbers (P < 0.001).” [2013] ” in some patients, complete elimination of certain bacterial communities.” [2004]
- “the beneficial commensal Faecalibacterium prausnitzii was sensitive to Essential Oils[Nerolidol, thymol, eugenol and geraniol] similar or even lower concentrations than the pathogens.” [2012]
- “Lactobacillus paracasei DG ..a concomitant decrease [2014]
- “The relative abundance of Faecalibacterium prausnitzii was unaffected except with thymol [Thyme Oil] at 500 p.p.m. of essential oils tested”
- “All strains tested were bile sensitive, showing at least 80% growth inhibition in the presence of 0.5 μg/ml bile salts, “[2012]
- Diet
- ” Faecalibacterium prausnitzii proportions decreased (P = 0.007, P = 0.031 and P = 0.009, respectively) as a result of the gluten-free diet” [2009]
- “was found to be significantly lower in broccoli-fed mice.” [2012]
- “the abundances of Bacteriodes, Prevotella, Peptostreptococcus and Faecalibacterium were lower in dogs fed the meat diet,” [2017]
- “Faecalibacterium prausnitzii… decreased significantly on the low-FODMAP diet as compared to baseline.” [2017]
- “enteral nutrition induced a further decline in diversity, as well as even lower levels of Faecalibacterium prausnitzii” [2016] [2014] [2014]
- “Malnutrition was associated to the reduction of several taxa, mainly related to the genus Bacteroides, Parabacteroides, Prevotella, Streptococcus, Faecalibacterium,” [2015]
- “High protein diet…associated with reduced concentrations of the Clostridium coccoides and C. leptum groups and Faecalibacterium prausnitzi” [2014]
- ” smokers had lower proportions of Faecalibacterium prausnitzii” [2016] [2012]
- “Oral Iron supplements treatment was associated with decreased abundances of operational taxonomic units assigned to the species Faecalibacterium prausnitzii,” [2017]
- “Commensal organisms (Bacteroides thetaiotaomicron VPI-5482 and Faecalibacterium prausnitzii A2-165) and a probiotic strain (Streptococcus thermophilus LMD-9) led to up to 12-fold induction of ferritin[iron] in colon.” [2016] — so taking iron supplements inhibits the bacteria that helps take up iron!
- Fiber
Update – Oct 2017
Since I wrote this, I discovered DataPunk.Net where a nice summary page is available: Faecalibacterium (genus)
NUTRIENTS/ SUBSTRATES (what it needs to live)
- Raffinose
- D-Glucose
- Galacturonic acid
- Riboflavin
- Arabinogalactan
- Stachyose (soy oligosaccharide)
- N-Acetyl-D-glucosamine
- Inulin
- Acetate
- Pectin
INHIBITED BY
- Chemotherapy
- High sugar diet
- High fat diet
- High animal protein diet
- Gallate
- Omega 3 fatty acids
- Fructo-oligosaccharides
- Bile salts
- Resistant starch (type IV)
- High animal fat diet
- Low fiber diet
- Low FODMAP diet
- Flaxseed
- High processed foods diet
- Navy bean (Cooked)
ENHANCED BY
- Fasting
- Epinephrine
- Resistant starch (type III)
- Jerusalem artichoke
- Saccharomyces boulardii
- Red wine
- Arabinogalactan
- Low animal protein diet
- Walnuts
- Low processed foods diet
- Chicory
- Inulin
- High fiber diet
- Berberine
- Dopamine
Bottom Line
In discussing this post with a friend, she said “WHAT! No Omega-3, No Iron, No gluten Free, No flaxseed, No essential oils, No Flagyl for yeast infections — and now you want me to put MSG on my food and eat tons of beans!!” The results of the review on PubMed had many surprises.
- Oral Iron (but not IV Iron) reduces the bacteria that absorbs iron – “you can’t get there from here“
- Fibre is not a magic general solution — it must be the right fibres and oligosaccharides — time to read the ingredients very carefully!
- A gluten free diet likely have lower amounts of the right fibres.
Some of the above fits common perception: No Smoking, no proton pump inhibitors, no antibiotics. For yeast infections use Rifaximin instead of Metronidazole (Flagyl) — there are studies finding it just as effective.
THIS IS NOT MEDICAL ADVICE — this post is an education summary of what has been reported on PubMed. Always consult with a knowledgeable medical professional before changing diet, supplements and prescription drugs.
To Physicians and the like: Stopping as many of the reducers as is practical and trying to alter the diet towards regular bean soups and de-alcholized red wine; with lingonberries for desert! If there are no histamines issue, Bacillus coagulans.