This is my 700th post and at the same time, a reader asked
Role of Official Medicine
I need to answer this based on my model and observations. Medicine is looking for simple cookbook solutions to a complex condition. Most practicing MDs are also greatly constrained by several nasty things:
- “Standards of Care” – “Practice guidelines assist the health care practitioner with patient care decisions about appropriate diagnostic, therapeutic, or other clinical procedures for specific clinical circumstances.” [NIH]
- “Medicare Insurance” – this is very closely tied to above
- “Peer pressure” – What would other doctors do in their region.
A professor once told me: “The ideal doctor or engineer is the person with a fantastic memory, strong sense of conformity and zero creativity”.
Give them a cookbook on how to treat an issue by a medical association, and they will follow it. Give them a ton of studies with conflicting results and they will either say “We will wait until there is clarification” or “It is probably psychological or best handled by a specialist”. Often the excuse can become “We need more studies”…. studies that may never happen. Most MDs do not want their rear side exposed to fellow MDs, local medical organizations and insurance companies for some very hard financial kicks.
Bottom Line: Official Medicine will stay in a holding pattern for years, likely decades. They will provide symptom relief and ad hoc random experiments (often repeating things tried unsuccessfully decades ago!). Only an extreme few would read any research on CFS regularly.
Research
My opinion based on seeing enough studies is that CFS/FM/IBS symptoms are interactions between DNA/SNP and microbiome (which has more DNA than a human). The appropriate research approach would be to take a large sample of patients (likely 100,000 or more) and record:
- DNA
- Microbiome
- Symptoms and Lab results
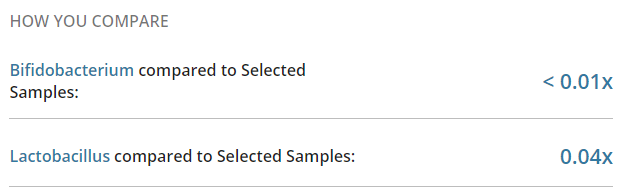
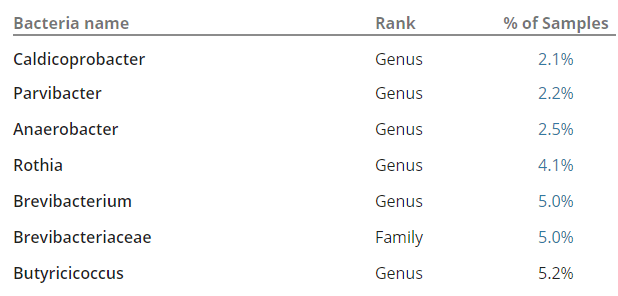
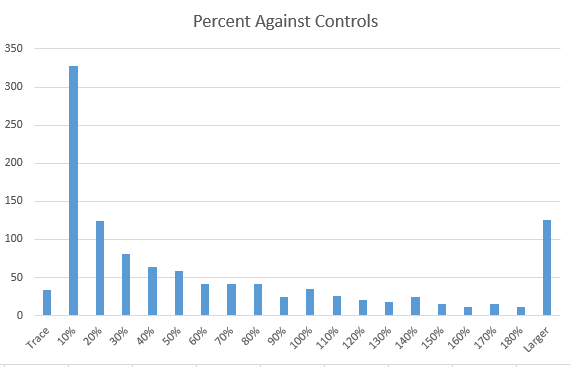
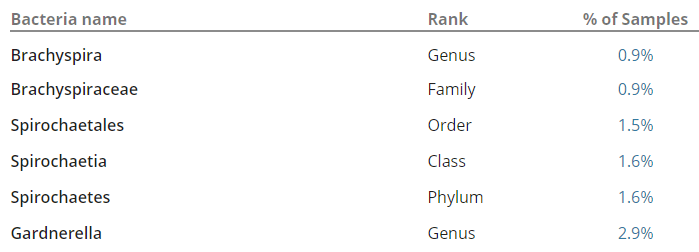
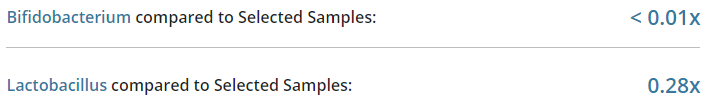
Then push all of this data in appropriate artificial intelligence algorithms to break down/discover the associations. In the comments of my last post, you would see that I identified several bacteria groups that are very high. When the reader tried any suggested herbs, they became severely depressed in days and stopped. A little research on PubMed discovered that most of these bacteria groups are strongly associated with depression. The die-off from the herbs was triggering the depression. One type of symptom==> a group of bacteria (and likely DNA also).
Current Researchers
Most researchers are trained in specialties. They do excellent work examining the leaves and twigs around their focus of interest. The problem with CFS is that the issue is bigger, not just the branches of the tree or the whole tree — rather the entire forest. IMHO, an ideal CFS researcher would be trained in medicine, DNA/SNP, microbiology and machine learning. They would need to be a world class data scientist besides being an MD. They will also need a big budget and cooperative clinics to get an adequate data sample (I am talking 100,000 patients needing their DNA and microbiome done).
To me, one of the greatest problems with CFS research is that successful results from studies are rarely repeated. My favorite soap box is pointing out the Berg’s studies finding a high (80-90%) of CFS patients having an inherited coagulation condition have never been repeated. Having a single study results in those results being ignored because “they have not been repeated” — ignoring the fact that no one tried to repeat them…
My Role
What is my role? Basically, it is as a recovered former patient who remembers the pain and hurt that this condition caused. I looked at my strengths and weaknesses and realized that my best service to the community is simple:
- Someone with a clear head who can read thru the literature, understand generally what the article actually says (i.e. cut thru researchers’ double speak). This arose from experience and training.
- I started reading medical articles and presentations at 14 years old because I was one of 15 students from my grade level in one of the largest school districts in Canada for a special enrichment program, Berg Science Seminars.
- I did an undergraduate and graduate degree in Statistics
- My Master’s topic was working with the Justice Institute of BC looking at how various medical treatments influenced survival during medical transport.
- I worked for many years as an Industrial Statistician and was a member of the American Statistical Association, as well as the American Mathematical Association, and a few other professional organizations.
- My day-to-day job deals with very complex systems and data – literally world class Information Technology Systems including Microsoft, Amazon and Starbucks. I am one of those few people who doesn’t suffer from information or complexity saturation, rather, I sit down and slowly shift and re-shift the information to build out and then test a model as to cause and solution. I have been (and still am) successful in this area. In July, 2017 I was the Edison Award Winner for the Starbucks Digital Platform Team.
My role is a citizen scientist modeler. It is “citizen” because I am not receiving any money for doing it. I have worked professionally as a scientist modeler.
My role is not:
- CFS Life Coach
- Action for getting more funding
- Social Support
- Treatment support
I could easily get involved with the above, but there are others who are more capable. My gifts are dealing with data and sketching out how the thousands of dots are connected. Taking these sketches and converting them into studies, treatment plans should be a role for the medical professionals. For example, Neem fits the model, but no one has done a clinical study on whether it is actually effective.
Bottom Line for Me
- Continue to review studies, cutting thru the medical double talk
- I am grateful for many readers who forward new studies to me for early review.
- Continue to test and refine the model that seems to fit best against all of the data: microbiome x DNA/SNP interactions
- Doing uBiome reviews help evolve the model
- Continue to identify items which may help — based on applying the model to known literature.
- Producing posts on these items with links to the research that I based it on (the keyword here is transparency)
- Advising patients that these items should be discussed with their medical professionals before starting. I have blinkers on and have no idea if the same item may cause a different medical condition to get worse. Only your medical professional can determine if it is safe to try or not. I do not want to get sucked into time-consuming understanding of interactions, I would rather spend that time connecting more dots.