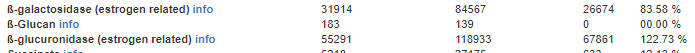While doing my monthly review of recent articles on PubMed dealing with the microbiome, I came across
- Alterations of Gut Microbiome in the Patients With Severe Fever With Thrombocytopenia Syndrome[2018].
- “Reduced gut microbiota diversity and dramatic shifts of fecal microbial composition in SFTS patients were observed compared with health controls. “
- “Lachnospiraceae and Ruminococcaceae which could produce short-chain fatty acids were clearly dropped. Sutterella which have anti-inflammation properties were reduced too. On the contrary, some common opportunistic pathogens like Enterococcus and Streptococcus and endotoxin-producing bacteria Escherichia which could rise the risk of infections were increased in SFTS patients than healthy people, in addition lactate-producing bacteria Lactobacillaceae also significantly increased in SFTS patients. “
- “the changes of gut microbiota of SFTS patients were closely associated with clinical symptoms”
This is in general what I expected to see with my model, especially due to the Bergen experience with a giardiasis outbreak. The increase of lactate producing bacteria is significant because with ME/CFS, a condition that often occurs post viral infection, is often associated with lactic acidosis.
“These results demonstrate that some patients have a disrupted microbiota following Norovirus infection, and therefore may be at elevated risk for long-term health complications.” [2012]
A third article, Antiviral effect of vitamin A on norovirus infection via modulation of the gut microbiome [2016]
- “We demonstrated the inhibitory effect of vitamin A against MNV replication both in vitro and in vivo. Interestingly, these inhibitory effects occurred directly or indirectly via microbiome changes, particularly on Lactobacillus strains in the gut…In previous studies, Lactobacillus spp. exhibited antiviral effects and alleviated the symptoms caused by rotavirus and influenza viral infections..In this study, we demonstrated that Lactobacillus spp. was enriched by vitamin A intake and significantly inhibited MNV replication. “
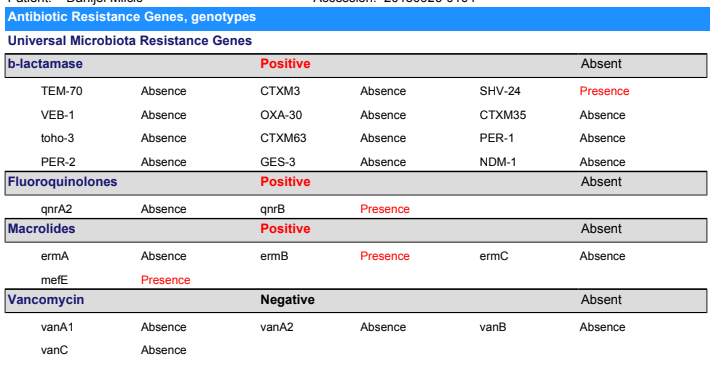
- “the abundance of Proteobacteria was increased by MNV inoculation, consistent with observations in patients whose gut microbiota was perturbed by human norovirus infection”
Bottom Line
A viral or bacterial infection may have significant impact on the microbiome. If the microbiome does not return to a healthy state, a wide variety of other conditions may arise in the days, months and years following.