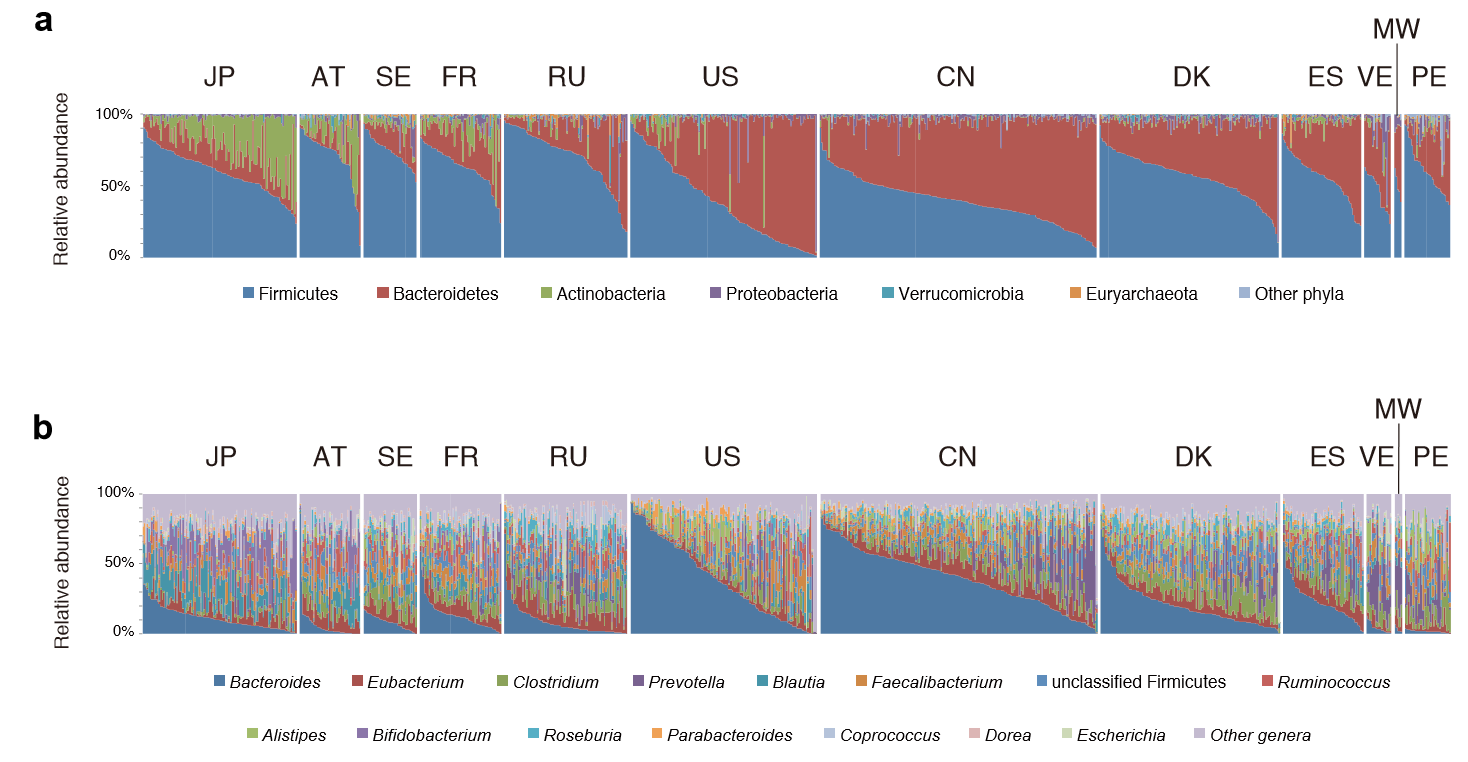
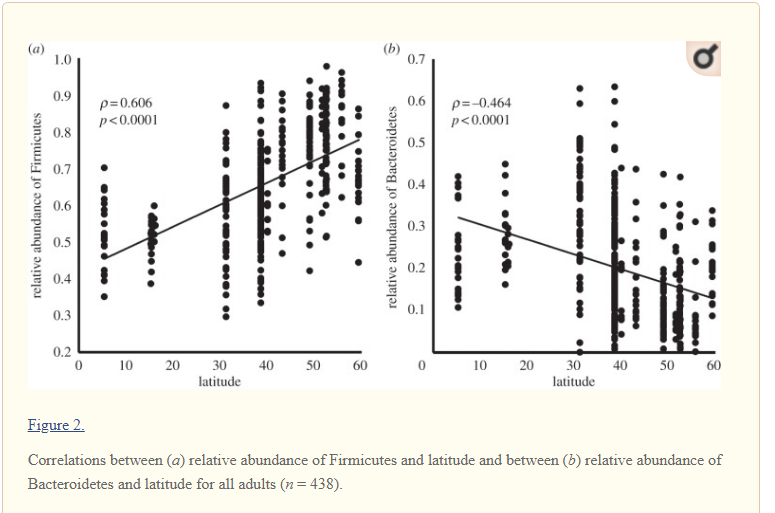
In an earlier post this week, I mentioned that a single patient microbiome can often be fuzzy to interpret. Regional lab reports where the patient is a match for the general population interms of genetics and diet is the exception. An example: Estonia and Sweden are a short boat ride away from each other but for L. Fermentum, this species was found only in Estonian children but not in Swedish children [2009].
A reader in Germany sent their result from a German Lab, GanzImmun Diagnostics AG.
The lab report has a nice presentation of how their results compared to a German reference population. The width of analysis was also good. Examples are below:
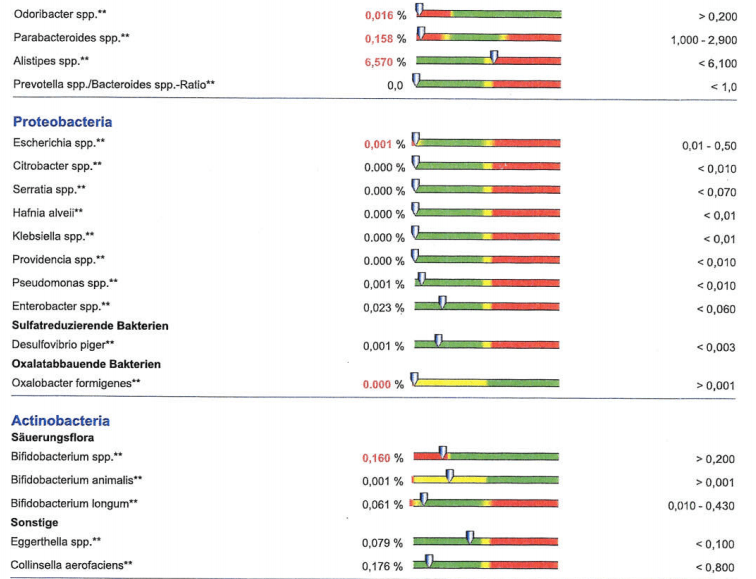

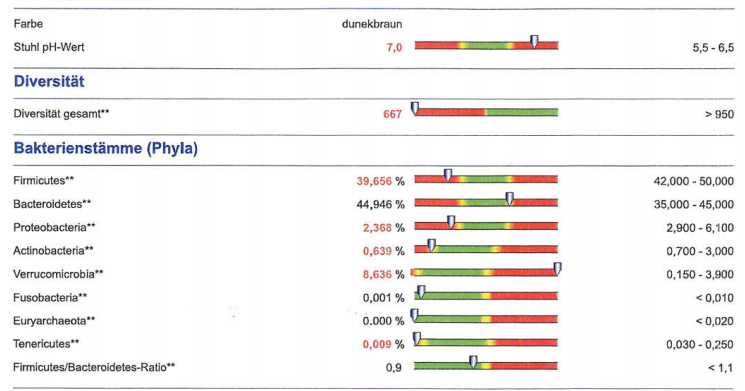


Compared to Other Reports
How does this compare to Lipkins results (for those reported above) and other results
- Faecalibacterium prausnitzii: Lipkins Low, Patient Low 🙂
- Roseburia: Lipkins Low, Patient Low 🙂
- Bacteroides vulgatus: Lipkins High, Patient Low 😦
- Dorea: Lipkins Low, Patient High 😦
And for the classic pattern reported in the 1998 Australian Study and others?
- Escherichia Coli etc: Study – Low, Patient – Low 🙂
- Bifidobacterium: Study – Low, Patient – Low 🙂
- Lactobacillus: Study – Low, Patient – Low 🙂
- Klebsiella/Enterbacter: Study – High, Patient – Normal 😐
- Enterococcus: Study – Low, Patient – Normal 😐
- Firmicutes: 2016 Study – Low, Patient – Low 🙂
- Bacteroides: 2016 Study – High, Patient – High 🙂
The model postulates that symptoms and microbiome shifts are likely associated. For the classic patterns, we have a good match. For Lipkin’s study, we have a much poorer match, causing the useful of that study further into question.
Checking for Infections
All negative, but an impressive list of items.

Additional Charts
Another chart was very interesting because it shows high histamine (Histaminbildner) producers — a frequent issue with CFS. Fäulnisflora (rotting flora) I suspect is SIBO, again common with CFS. Säuerungsflora(acidifying bacteria) is low And high pH (alkaline) as the consequence.
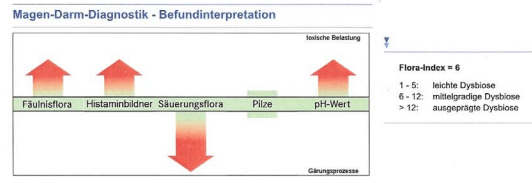
Bottom Line
The first thing that struck me was to reduce pH. A shift in pH results impacts a lot of bacteria (increase or decreases growth).
pH Factors
Normal pH is 5.5 to 6.5, this patient is 7.0. A shift outside of the normal range is usually viewed as an indicator of bacteremia [2012]. Based on Bile Acids: A pH Dependent Antibacterial System in the Gut? [BMJ 1972], bile acid supplementation should be discussed with your medical professional. Drinking diluted vinegarhas often been suggested on alternative-medicine sites. For myself, I prefer eating pickled herring – Bismarckhering because it is natually rich in all of the recommended supplementations for CFS
- “Pickled herring is one of the best sources of natural vitamin D3. It is also an excellent source of selenium and vitamin B12. 100 grams may provide 680 IU of vitamin D or 170% of the DV, as well as 84% of the DV for selenium and 71% of the DV for vitamin B12. [5][6]” – Wikipedia
- “Increasing levels of the primary bile acid cholic acid (CA) causes a dramatic shift toward the Firmicutes, particularly Clostridium cluster XIVa” [2013][2017]
- Western diet deregulates bile acid homeostasis, cell proliferation and tumorigenesis in colon [2017]. “Western-style diets (WD) high in fat and scarce in fiber and vitamin D”
- “Pickled herring is one of the best sources of natural vitamin D3. It is also an excellent source of selenium and vitamin B12. 100 grams may provide 680 IU of vitamin D or 170% of the DV, as well as 84% of the DV for selenium and 71% of the DV for vitamin B12. [5][6]” – Wikipedia
I will revisit pH (Acid/Alkaline balance) in a future post — there are some complexities (fecal pH versus blood pH) and few studies to reference. A high stool pH is seen with non-specific diarrhea, crohn’s disease, ulcertative colitis, ileostomy [1970], the general spectrum of autoimmune conditions that CFS patients could drift into over time. Giadia infections (cause of a CFS outbreak in Norway) appears to result in an increase of pH [2001] — suggesting a possible mechanism for CFS onset. The triggering event caused a pH shift which resulted in a change of bacteria. The change of bacteria seized the opportunity to keep the pH favorable to these bacteria. For example, ammonia producing bacteria would cause pH to increase.
- “A shift to an alkaline environment ..is stressful for bacteria as shown by how Escherichia coli responds to alkali with SOS and heat shock-like responses [18]; [19] ; [20].”
- “B. subtilis initially grows rapidly and acidifies the medium.” [2005]
“An increase in the relative abundance of Bacteroides spp., Blautia spp., Parabacteroides spp., Coprococcus spp., Ruminococcus spp., and Oscillospira spp. and a decrease of Bifidobacterium spp., Lactobacillus spp., Escherichia spp., and Clostridium spp. were observed during weaning. The change in microbiome composition was accompanied by a gradual increase of fecal pH from 5.5 to 7.” [2016] That is, the result from going from human milk to cow milk in humans. Human milk have a pH of 7.4 [1986] while cow milk has a pH of 6.5 [source] and milk from different mammals are significantly different in composition [2004]

Antibiotics
The reader asked about antibiotics. I have done some very rough charts on antibiotics and what they work against. The general group that this patient is high in are Bacteroides spp, with specific members being low — adding some complexities. Using Trends in antimicrobial resistance among Bacteroides species and Parabacteroides species in the United States from 2010-2012 with comparison to 2008-2009 [2017].Which suggest metronidazole or chloramphenicol or Carbapenem or beta-lactam/beta-lactamase inhibitor combinations. Based on the CFS patients survey on response to antibiotics, I would opt for metronidazole(a.k.a. Flagyl) as the first choice.
Bottom Line
Doing this analysis, I came across the pH aspect of CFS and will do further digging in the literature. My suggestions to discuss with a medical professional are:
- Regular intake of food that are high in acid (which is different from food causing an “acid stomach”)
- Metronizalole
- Bacillus probiotics, see this post — some may not be suitable for everyone
- “Cultures of B. subtilis were popular worldwide before the introduction of antibiotics as an immunostimulatory agent to aid treatment of gastrointestinal and urinary tract diseases” [wikipedia]