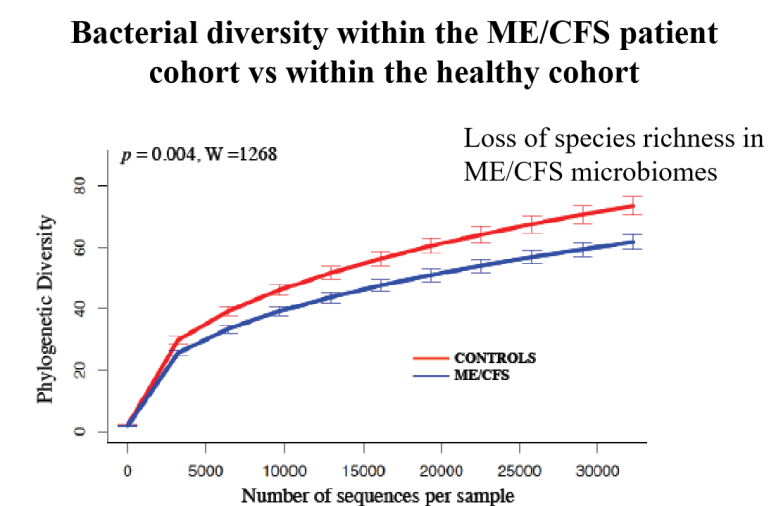
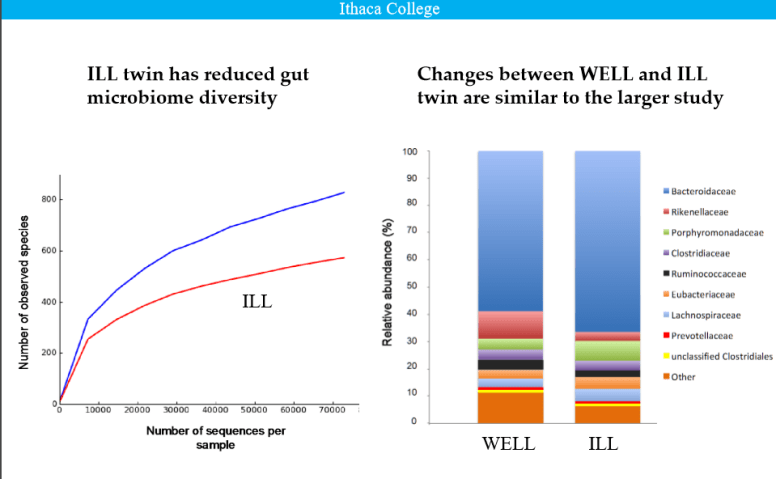
I have known chronic fatigue syndrome (CFS) patients that prior to CFS had no problem with lactose. As CFS progressed they became intolerant.
In reviewing PubMed Studies, I found that CFS patients with lactose intolerance was excluded from some studies of CFS [2002]. Other papers report overlap of IBS and LI [2005]. A 2011 study summarized it nicely “Substantial overlap of symptoms and comorbidities occur not only between irritable bowel syndrome and other functional gastrointestinal disorders but also with gastrointestinal disorders that are not related to motility (eg, celiac disease and lactose intolerance) and to somatic conditions (eg, fibromyalgia and chronic fatigue syndrome).” [2011]
Lactose intolerance as a result of a persistent shift of gut bacteria appears consistent.
“Lactose malabsorption and milk products intolerance symptoms are the most common alimentary tract disorders. Lactose intolerance is a result of lactase deficiency or lack of lactase and lactose malabsorption. Three types of lactase deficiency were distinguished:
- congenital,
- late-onset lactase deficiency and
- secondary lactase deficiency.
Lactose intolerance means the appearance of clinical gastrointestinal symptoms after ingestion of lactose. To the clinical symptoms of lactose intolerance belongs: nausea, vomiting, abdominal distension, cramps, flatulence, flatus, diarrhea and abdominal pain.” [2009]
“All the patients with lactase deficiency(LD) were detected to have small intestinal (SI) bacterial overgrowth (BOG). An inverse correlation was found between LD and the degree of SI BOG (r = -0.53; p < 0.001). 73.7% of the patients with moderate LD showed a positive effect of probiotic therapy ….. No improvement occurred in 73.8% of the patients with severe LD.”[2015]
“secondary lactase deficiency (SLD) was detected in 59.4% of patients with postinfectious IBS [i.e. IBS resulted from an infection]. Mild SLD was determined in 43.5% of patients, and severe SLD – in 15.9% of patients.” [2012]
- “lactase deficiency (LD) was identified in 36.5% of the patients with PI-IBS. There was an inverse correlation between the degree of LD and SI BOG. The good therapeutic effect of probiotics in LD suggests that the symbiotic gut microflora positively affects the activity of lactase in the human SIM. No therapeutic effect of probiotics in patients with severe LN serves as the basis for a search for more active probiotic therapy.” [2015]
“The majority of people with lactose malabsorption do not have clinical lactose intolerance. Many individuals who think they are lactose intolerant are not lactose malabsorbers.” [2010]
“A recent meta-analysis permitted to show that almost all lactose intolerants tolerate 12 g of lactose in one intake and approximately 18 g of lactose spread over the day.” [2015]
“In spite of public knowledge and advertising, controlled studies did not prove the beneficial effect of either a lactose-free diet, enzyme supplementation or probiotics in an evidence-based manner.” [2015]
” Most individuals with LI can tolerate up to 12 grams of lactose, though symptoms became more prominent at doses above 12 grams and appreciable after 24 grams of lactose; 50 grams induced symptoms in the vast majority. A daily divided dose of 24 grams was generally tolerated. We found insufficient evidence that use of lactose reduced solution/milk, with lactose content of 0-2 grams, compared to a lactose dose of greater than 12 grams, reduced symptoms of lactose intolerance. Evidence was insufficient for probiotics (eight RCTs), colonic adaptation (two RCTs) or varying lactose doses (three RCTs) or other agents (one RCT). Inclusion criteria, interventions, and outcomes were variable. Yogurt and probiotic types studied were variable and results either showed no difference in symptom scores or small differences in symptoms that may be of low clinical relevance.” [2010]
A 2004 article recommends DNA testing “Attention should be paid to appropriate interpretation of genetic detection in order to avoid potentially harmful reduction in dairy intake or misdiagnosis of secondary lactase deficiency.” ” These are C/T 13910 and G/A 22018 substitutions.”[2009] “The LCT(T/C-13910) polymorphism is associated with subjective milk intolerance, reduced milk calcium intake,” [2004]
Treatment
- ” The results indicated a definitive change in the fecal microbiome of lactose intolerant individuals that were clinically responsive to dietary adaptation to short chain galacto-oligosaccharide (GOS), named RP-G28″[2013]
- ” in 1996, 20 people, with various ethic backgrounds, and who were all poor at digesting lactose, were randomly assigned to take either a small dose of lactose or one of dextrose, a different sugar, for 10 days… Encouraging, the course of small lactose doses did improve tolerance: after the 10-day course of lactose,” [1996]
- Thus complete avoidance as was advocated for peanuts (the common belief) is likely not ideal
- ” In the absence of guidelines, the common therapeutic approach tends to exclude milk and dairy products from the diet. However, this strategy may have serious nutritional disadvantages. Several studies have been carried out to find alternative approaches, such as exogenous beta-galactosidase, yogurt and probiotics for their bacterial lactase activity, strategies that can prolong contact time between enzyme and substrate delaying gastrointestinal transit time, and chronic lactose ingestion to enhance colonic adaptation.” [2009]
- The effects of the DDS-1 strain of lactobacillus on symptomatic relief for lactose intolerance – a randomized, double-blind, placebo-controlled, crossover clinical trial [2016]. “The DDS-1 strain of Lactobacillus acidophilus, discovered in 1959 by Dr. Khem Shahani at the University of Nebraska, is a unique strain of L. acidophilus on deposit with the FDA Agricultural Research Service (ARS) with the catalog number B-3208. It is currently manufactured by Nebraska Cultures, Inc. A recent study demonstrated the DDS-1 strain of L. acidophilus to be superior to other strains of lactobacillus in the ability to establish in the human gastrointestinal (GI) tract [43]…can provide symptom benefit compared with placebo among individuals who consume the product for a course of 4 weeks.”
- ” Treatment of lactose-maldigesting subjects with and without hypochlorhydria with Lactobacillus acidophilus BG2FO4 for 7 d failed to change breath-hydrogen excretion significantly after lactose ingestion.” [1999] – so it is strain specific
- “In lactose intolerants, tilactase strongly improves both LBT results and gastrointestinal symptoms after lactose ingestion with respect to placebo. Lactobacillus reuteri also is effective but lesser than tilactase.” [2010]
- “Feeding yogurt that was pasteurized following fermentation, with only trace amounts of microbial beta-galactosidase activity, results in a threefold increase in lactose malabsorption as compared with feeding yogurt with a viable culture.” [1987]
- “There is a poor correlation between lactose maldigestion and intolerance; in some studies,” [2001]
- Beneficial effects of long-term consumption of a probiotic combination of Lactobacillus casei Shirota and Bifidobacterium breve Yakult may persist after suspension of therapy in lactose-intolerant patients [2012]. “Four-week consumption of a probiotic combination of L casei Shirota and B breve Yakult seems to improve symptoms and decrease hydrogen production intake in lactose-intolerant patients. These effects may persist for at least 3 months after suspension of probiotic consumption.”
- “Sixty patients with moderate secondary lactase deficiency (SLD) were randomized to 2 groups: 1) 41 patients received basic therapy (mesim forte as one tablet t.i.d., no-spa, 40 mg, t.i.d.) and combined probiotic bifiform (Ferrosan) containing Bifidobacterium longum 107, Enterococcus faecium 107 as one capsule t.i.d. for 14 days….After a 14-day course of therapy with the combined probiotic bifiform, restoration of eubiosis in the small bowel lumen was achieved in 70.8% of the patients,” [2013]
- “the probiotic Bifiform, composed of Bifidobacterium longum 10(7) and Enterococcus faecium 10(7), demonstrated efficiency in correction of mild SLD in patients with postinfectious IBS and can be used to prevent SIBO.” [2012]
Vitamin D and LI
- A relationship between vitamin D, parathyroid hormone, calcium levels and lactose intolerance in type 2 diabetic patients and healthy subjects [2016]. “levels of plasma 25-OH vitamin D (total), parathyroid hormone and serum calcium were more decreased in lactose intolerant diabetic patients than lactose tolerant patients. Sixty seven percent (67%) of diabetic patients suffered from osteoporosis and 20% of controls. Eighty percent (80%) diabetic patients and 16% controls with osteoporosis suffered from lactose intolerance.”
Bottom Line
There is weak evidence that Lactobacillus Reuteri, Lactobacillus casei Shirota (Yakult), Bifidobacterium Breve and explicitly Lactobacillus acidophilus DDS-1 may reduce LI but will make CFS worst (increase TNF-alpha even higher). Ferrosan Bifiform has stronger evidence. It is surprising that relative few probiotics have been tested. The association with IBS suggests that IBS probiotics may also reduce LI: Prescript Assist, Mutaflor, Symbioflor-2, Align.
Vitamin D levels may play a role, and thus supplementation of 10-15000 IU of vitamin D should be discussed with your medical professional.
Complete lactose avoidance appears to be unnecessary, and may actually make LI worst.
Lactobacillus acidophilus DDS-1 is available as a single probiotic – read this post on it,