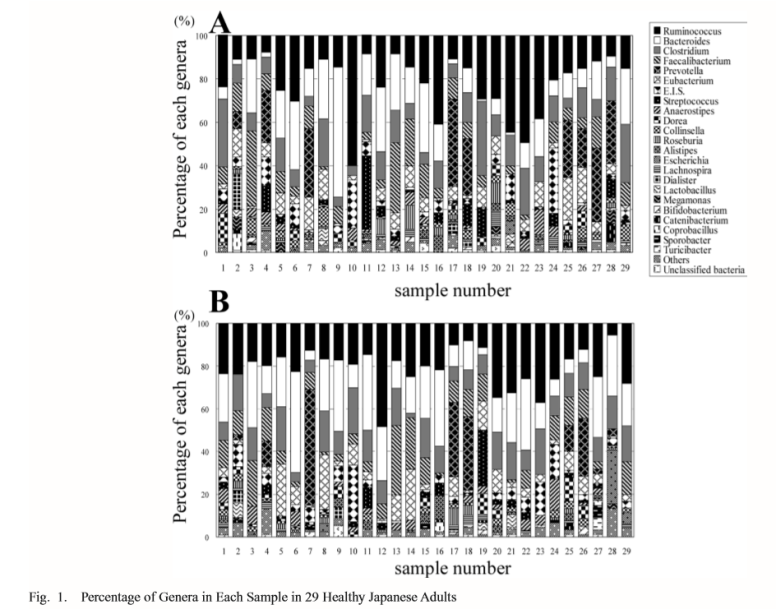
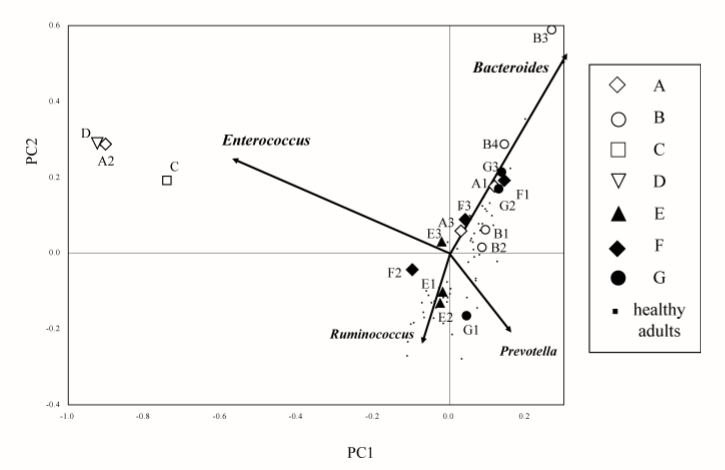
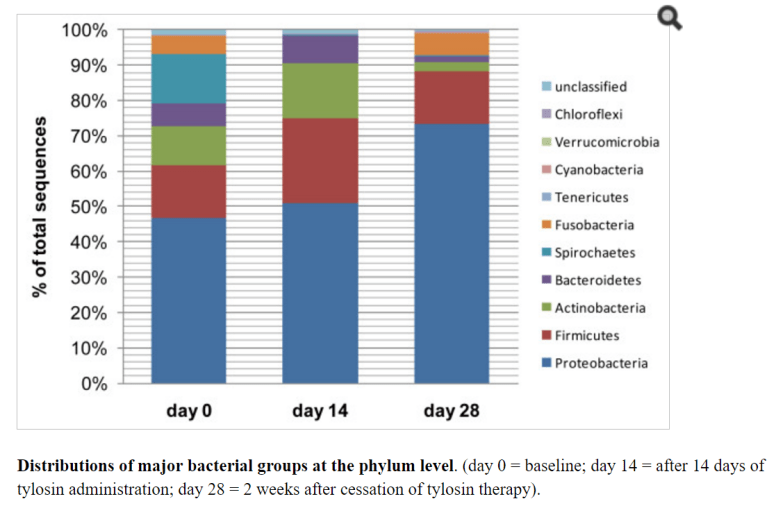
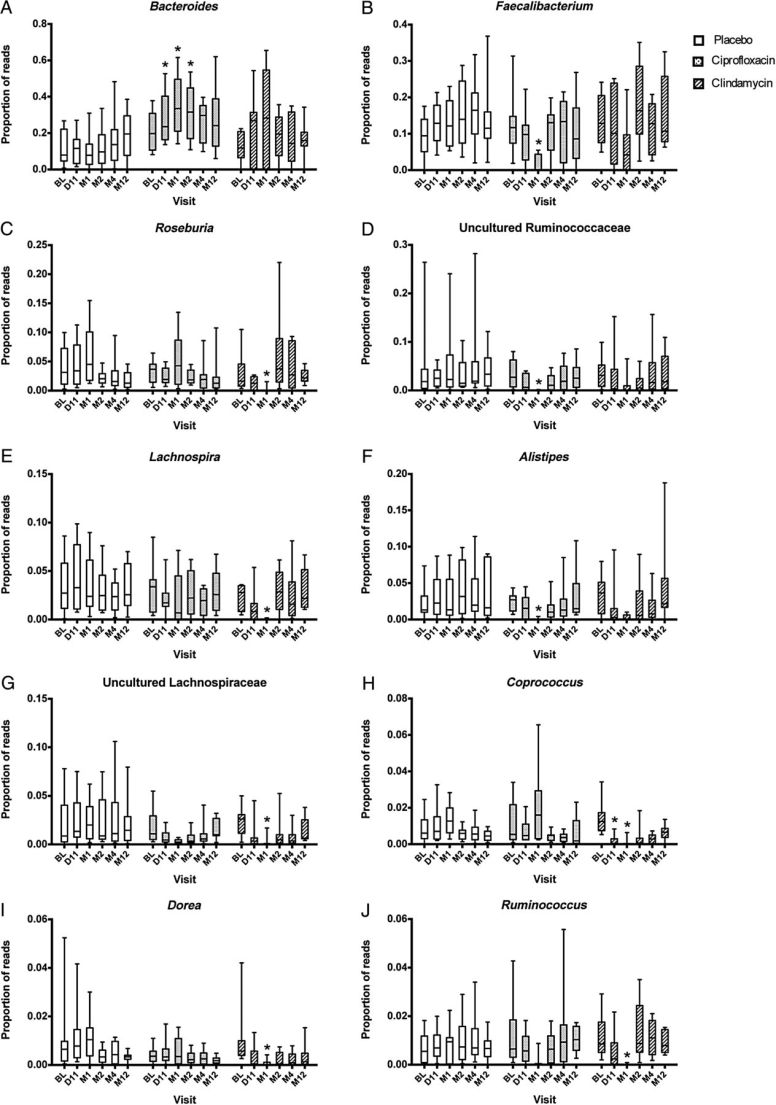
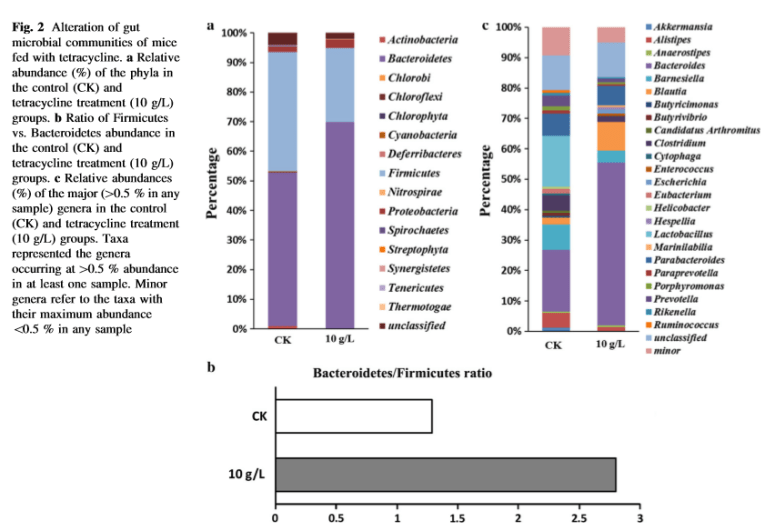
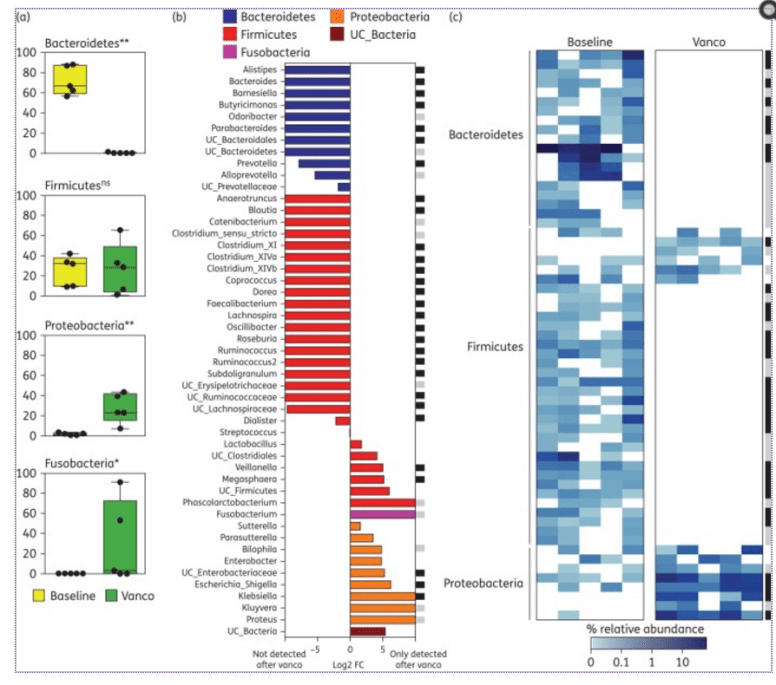
This issue often comes up in discussions with CFS/FM/IBS
- “Recent research shows that patients with CFS have marked alterations in microbial flora, including lowered levels of bifidobacteria and small intestinal bacterial overgrowth (SIBO).” [2003]
- “a considerable proportion of patients with fibromyalgia experience gastrointestinal symptoms that are commonly overlooked in the studies that are not specifically dedicated to evaluate these manifestations.” [2015]
- “Recent investigations have shown that bacterial overgrowth of the small intestine is associated with a number of functional somatic disorders, including irritable bowel syndrome (IBS), fibromyalgia, and chronic fatigue syndrome.” [2002]
The model that I use is based on a shift of the microbiome — some overgrowth and some undergrowth. SIBO is a subset of this definition.
According to wikipedia, there are multiple methods of diagnosis — the basis of these methods is abnormal results with “some physicians suggest that if the suspicion of bacterial overgrowth is high enough, the best diagnostic test is a trial of treatment. If the symptoms improve, an empiric diagnosis of bacterial overgrowth can be made.[39]“
Under treatment: ” However, if the condition recurs, antibiotics can be given in a cyclical fashion in order to prevent tolerance. For example, antibiotics may be given for a week, followed by three weeks off antibiotics, followed by another week of treatment. Alternatively, the choice of antibiotic used can be cycled.[39]” — the cycling of antibiotic choices is a regular part of Jadin’s protocol for CFS.
Latest from PubMed
- For rifaximin, “The overall eradication rate according to intention-to-treat analysis was 70.8%…The overall rate of adverse events was 4.6%… improvement or resolution of symptoms in patients with eradicated SIBO was found to be 67.7%”[2017]
- “Although the rifaximin group showed a greater percentage of global symptom improvement, this was limited to bloating, as scores for abdominal pain, diarrhea, and constipation did not improve significantly.”[2007]
- “SIBO was found to be associated with deep vein thrombosis (DVT).” [2016] — which implies coagulation may be associated with SIBO (i.e. thick blood).
Bottom Line
SIBO is a common condition with vague diagnosis criteria. Often the treatment and diagnosis is for one particular group of bacteria — for example Hydrogen Sulfide. A MD may tell the patient, your SIBO is resolved and the reality is that only the Hydrogen Sulfide producing bacteria has been reduced, the rest remain active — creating symptoms of FM, CFS, etc.
In short, SIBO — both tests and treatment are a part of the solution to CFS/FM/IBS but is not the full answer and likely, IMHO, not a major part of the answer. False positives and negatives are common with common tests.
On the PLUS SIDE — as with chronic lyme, SIBO can be a justification(defense) for a MD to prescribe antibiotics. Prescribing antibiotics for CFS or FM is not politically acceptable in most countries.