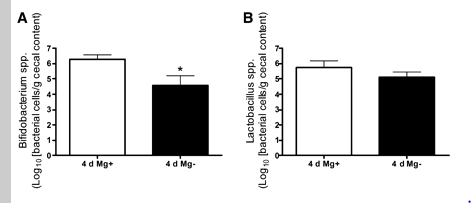
In my last post we found that magnesium enhanced the growth of bifidobacteria and lactobacillus — two groups of bacteria that CFS patients are low in. The majority of PubMed studies found that it also improve symptoms. Yet magnesium levels were found to be normal in blood cells — which raise the issue of “selective rationing of minerals and vitamins to the most critical components”. The blood is far more critical to maintain magnesium levels than incidental bacteria in the gut. Often testing assumes that the body is naive and does not do selective rationing.
In this next re-examination of supplements shown to help CFS/FM/IBS, I will look at Vitamin D. I have written often about the need to take sufficients (Mythology of Vitamin D toxicity, changes from increasing Vitamin D, Vitamin 25D and 1,25D). A few citations:
- “This study demonstrates for the first time a direct antiviral effect of vitamin D in an in vitro infectious virus production system.”[2011]
- “vitamin D is the environmental factor that most strongly influences autoimmune disease development.”[2015]
- “A significant negative correlation between vitamin D level and widespread pain index was found.”[2012] i.e. FM
- Serum 25-Hydroxyvitamin D3 and BAFF Levels Are Associated with Disease Activity in Primary Sjogren’s Syndrome [2017]. “Female SS patients had significantly lower vit D levels than controls” [2015]
- “Our findings showed that the high-dose supplementation of vitamin D[9 of 50000 IU cholecalciferol capsules for 3 months taken at weekly intervals] affects measures of systemic inflammation: reductions in High-Sensitivity C-Reactive Protein level and Neutrophil-to-lymphocyte ratio (NLR) distribution.” [2017]
- “The results of the meta-analysis of 10 trials involving a total of 924 participants showed that vitamin D supplementation significantly decreased the circulating hs-CRP level by 1.08 mg/L” [2014]
A longer quote from this year is beneficial
“Vitamin D was first discovered as the curative agent for nutritional c, and its classical actions are associated with calcium absorption and bone health. However, vitamin D exhibits a number of extra-skeletal effects, particularly in innate immunity. Notably, it stimulates production of pattern recognition receptors, anti-microbial peptides, and cytokines, which are at the forefront of innate immune responses. They play a role in sensing the microbiota, in preventing excessive bacterial overgrowth, and complement the actions of vitamin D signaling in enhancing intestinal barrier function. Vitamin D also favours tolerogenic rather than inflammogenic T cell differentiation and function. Compromised innate immune function and overactive adaptive immunity, as well as defective intestinal barrier function, have been associated with IBD. Importantly, observational and intervention studies support a beneficial role of vitamin D supplementation in patients with Crohn’s disease, a form of IBD. This review summarizes the effects of vitamin D signaling on barrier integrity and innate and adaptive immunity in the gut, as well as on microbial load and composition. Collectively, studies to date reveal that vitamin D signaling has widespread effects on gut homeostasis, and provide a mechanistic basis for potential therapeutic benefit of vitamin D supplementation in IBD.” [2017]
Now for some interesting links:
- Oral supplementation with probiotic L. reuteri NCIMB 30242 increases mean circulating 25-hydroxyvitamin D: a post hoc analysis of a randomized controlled trial. L.Reuteri is low in CFS patients, thus a decrease would be expected.
- Vitamin D Promotes Pneumococcal Killing and Modulates Inflammatory Responses in Primary Human Neutrophils [2017].
- “Vitamin D intake .. Prevotella was more abundant .., while Haemophilus and Veillonella were less abundant … and abundances of Coprococcus and Bifdobacterium … were inversely correlated with 25(OH)D.”[2017] in other words, Vitamin D alters the microbiome.
- “As a result of these effects on the intestinal mucosa, maintenance of sufficient vitamin D status may be essential for the development of a healthy gut microbiota, particularly in conditions defined by chronic mucosal inflammation such as CF…additional evidence is needed to establish vitamin D as a therapeutic approach for gut microbiota modification.” [2016]
- Vitamin D deficiency changes the intestinal microbiome reducing B vitamin production in the gut. The resulting lack of pantothenic acid adversely affects the immune system, producing a “pro-inflammatory” state associated with atherosclerosis and autoimmunity[2016]. “1) Seasonal fluctuations in vitamin D levels have normally produced changes in the intestinal microbiome that promoted weight gain in winter. Years of vitamin D deficiency, however, results in a permanently altered intestinal environment that no longer favors the “healthy foursome”. 2) Humans have always had a commensal relationship with their intestinal microbiome. We supplied them vitamin D, they supplied us B vitamins. 3) The four species that make up the normal microbiome are also commensal, each excretes at least one B vitamin that the other three need but cannot make. 4) Improved sleep and more cellular repairs eventually depletes body stores of pantothenic acid, causing reduced cortisol production, increased arthritic pain and widespread “pro-inflammatory” effects on the immune system. 5) Pantothenic acid deficiency also decreases available acetylcholine, the neurotransmitter used by the parasympathetic nervous system.”
Bottom Line
The understanding of what Vitamin D does have changed over the last decades. Originally, it was know to cure rickets (and the recommended dosage became some 50 Iu/day), then it impacted bone loss and the dosage increased. Today, we find that it is implicated in many areas, impacting the distribution of bacteria in the microbiome, and needed for a healthy gut microbiota.
The biggest problem is that MDs are often behind the times and do not work from the latest literature. For example a single dosage of 600,000 IU of vitamin D is deemed safe on medscape, or 10,000 IU/day for months [source]. One of the studies above cited an average of 60,000 IU/day for 3 months! [2017]