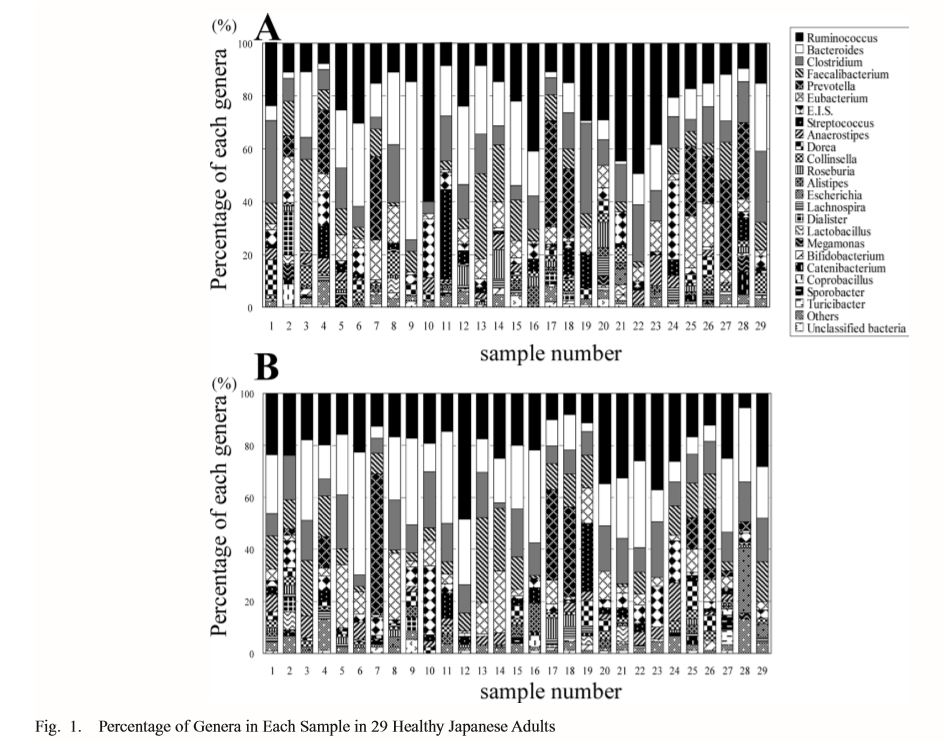
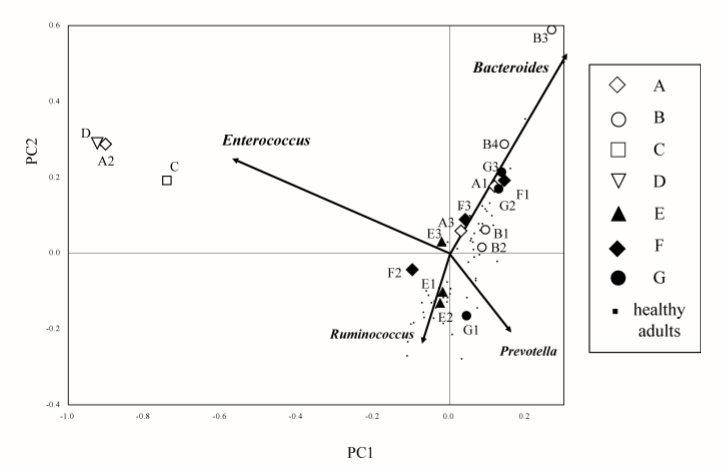
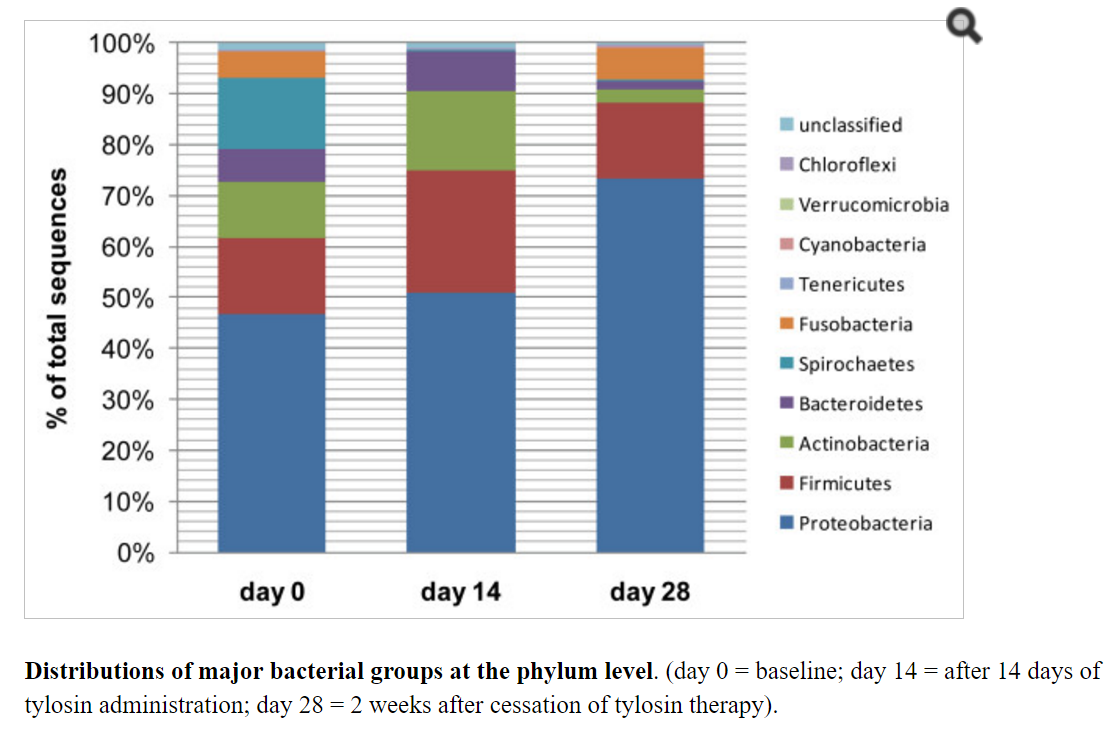
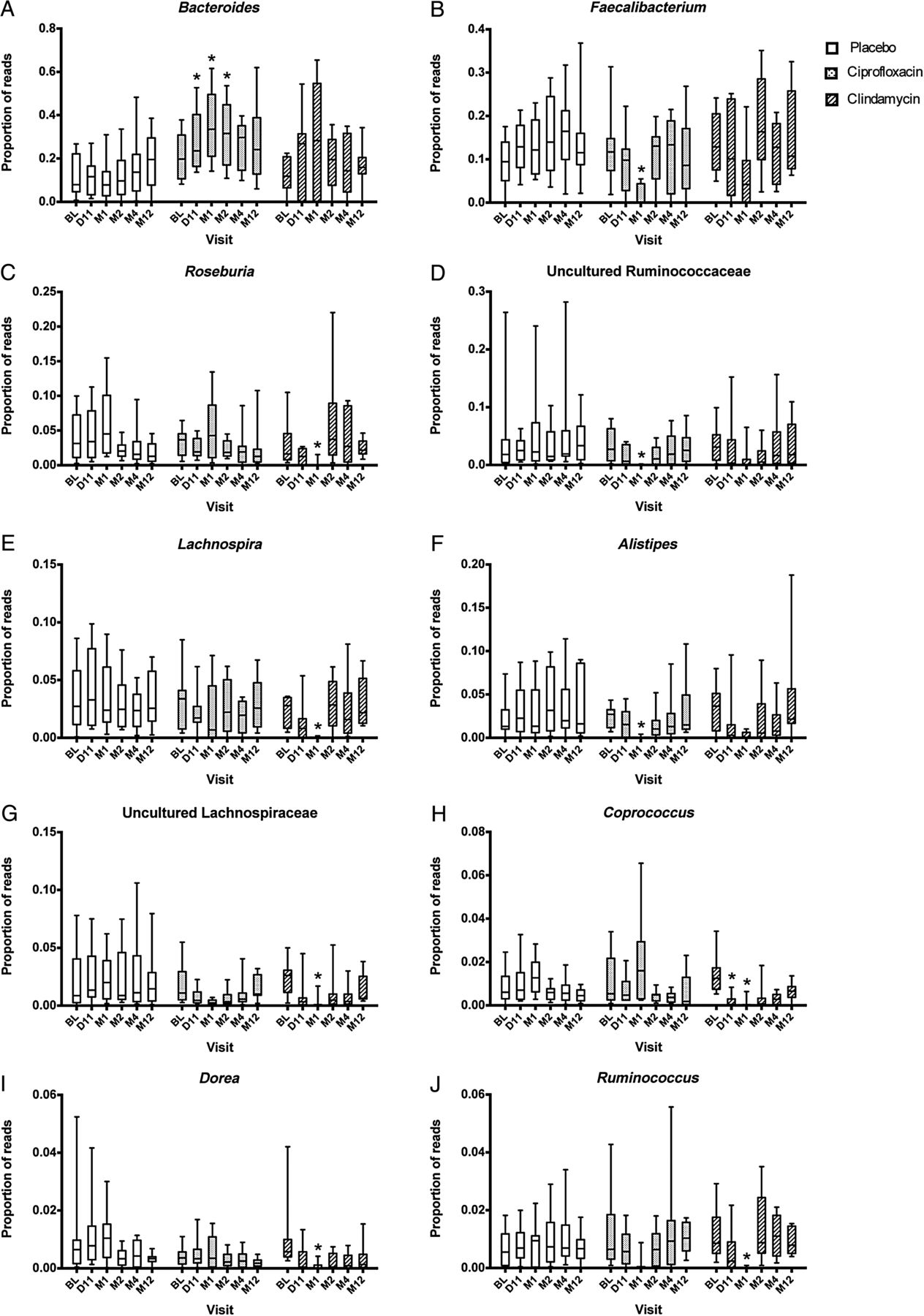
The next family of antibiotics that I found studies on was penicillin, typically amoxicillin. With many older theories of CFS, this family of antibiotics was believed to encourage the growth of bacteria suspected to cause CFS. Often you will read of bad reactions of CFS patients to this family of antibiotics. There appear to be some “truth” to this — but in a negative sense — it does not encourage the growth — rather it reduces already low level of two major families of bacteria seen with CFS patients… so yes, it will likely make most CFS patients worst.
“Oral amoxicillin exposure caused marked shifts in microbiome composition that lasted approximately 30 days on average and were observed for more than 2 months in some of the treated individuals .” [2016]
- “The use of amoxicillin also influenced the intestinal microbiota, significantly decreasing the CFU of Bifidobacterium spp. and Lactobacillus spp./g of feces.” [2005]
The first study was interesting Prospective randomized controlled study on the effects of Saccharomyces boulardii CNCM I-745 andamoxicillin-clavulanate or the combination on the gut microbiota of healthy volunteers[2016].

Unfortunately the rest of the images in this study were well above the cognitive level of most CFS patients.
Microbial diversity in individuals and their household contacts following typical antibiotic courses [2016]. “As few as 3 days of treatment with the most commonly prescribed antibiotics can result in sustained reductions in microbiota diversity,” Again, this article have complex charts.
Long-term changes in human colonic Bifidobacterium populations induced by a 5-day oral amoxicillin-clavulanic acid treatment [2012]. is of special interest becasue bifidobacterium is low with CFS patients. This antibiotic makes it worst!

I have created a page that processes existing literature and produces a list of suggestions on what may counteract the antibiotic shift. The page is [here]