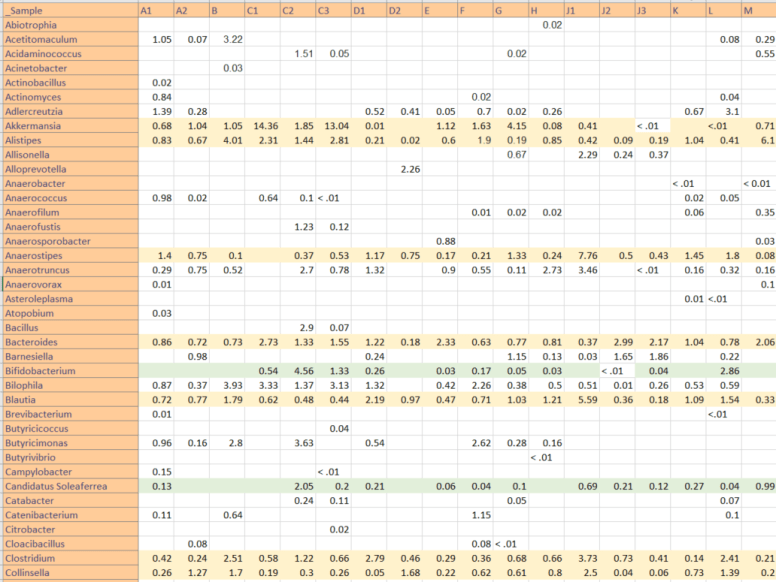
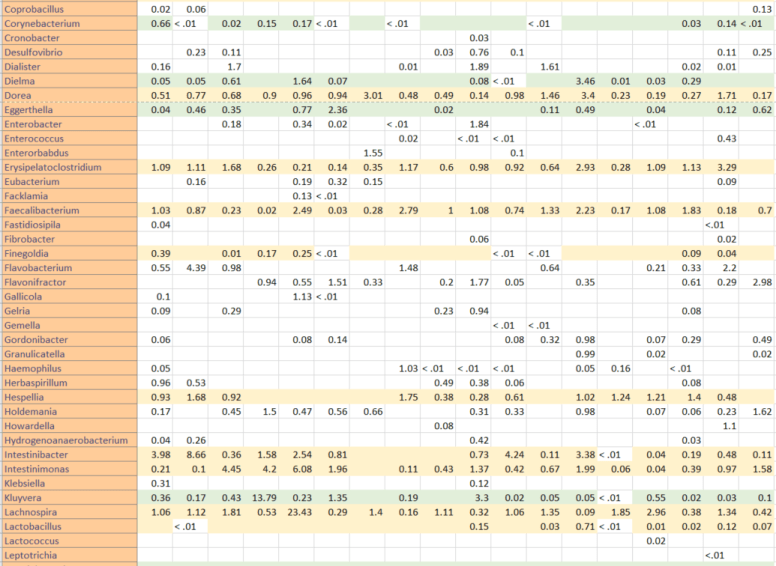
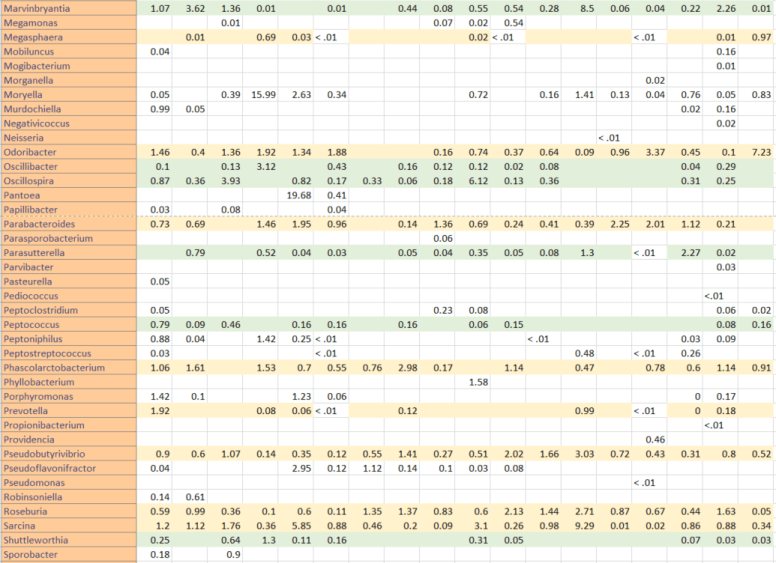
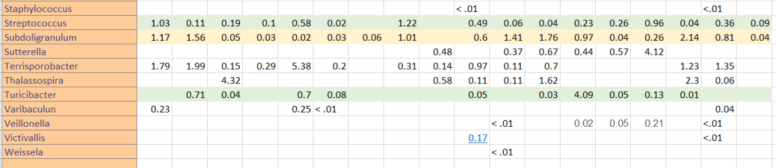
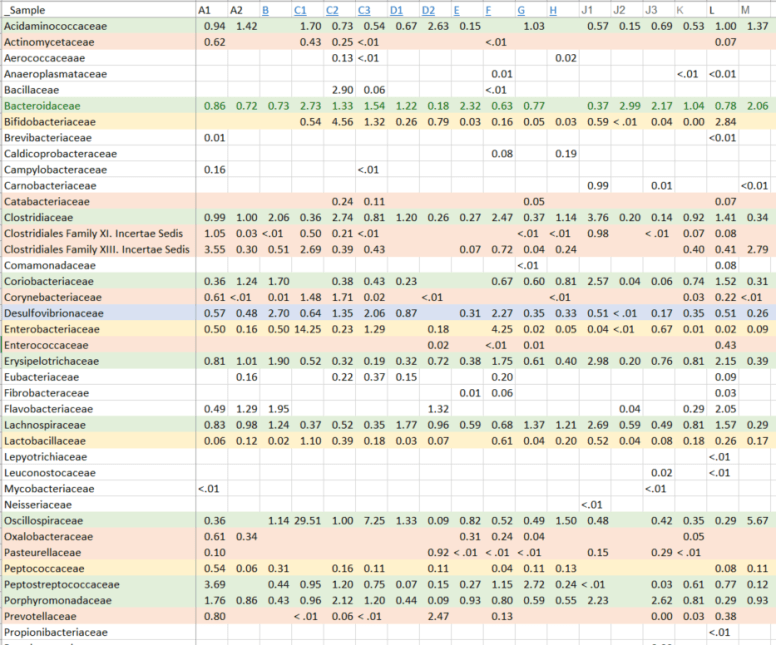
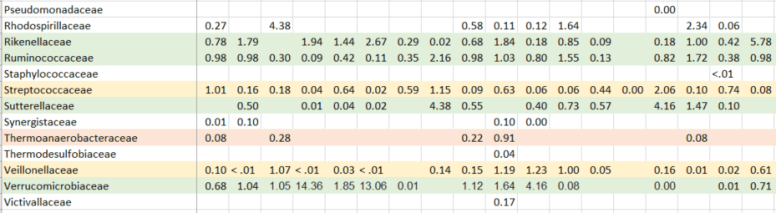
Since I started this blog, some 530 posts ago, there have been significant changes in probiotics available and information available (for example uBiome results). My original model favored the reports from the University of Newcastle in Australia in 1998. The uBiome results from a sample of over a dozen readers of this blog confirmed parts of this report and also raise questions on other parts. In re-reading the report, there was only 4 controls to 27 patients — which may account for the differences they saw. I summarized below:
| 1998 Study | uBiome Sample | |||
| Controls | CFS | Ratio | Ratio | |
| Bifidobacterium | 7% | 2% | 28% | 21% |
| Bacteroides | 93% | 91% | 98% | 98% |
| Enterococcus | 1% | 24% | 2400% | 0% |
| Enterobacter | 0% | 3% | 30000% | 18% |
| E.Coli | 92% | 49% | 53% | 18% (parents) |
In short, we do not see the overgrowth, but can confirm the undergrowth matches from the uBiome results.
There was a match up of the antibiotics used by Jadin with the overgrowth and undergrowths, which reinforced the model. There may a different reason that her antibiotics protocol works for many — for example, inhibiting “normal” bacteria allowing the undergrowth to increase. I do not have clarity on this.
My rotate rotate rotate was based on the belief that there was there was significant overgrowth that could become resistant. I have now diminish it’s importance (I have not abandoned it).
Suggestions for an updated Probiotic Protocol
All of the above resulted in my recommendations to discuss with your medical professional:
- Start with General Biotics Equilibrium – you will keep taking it while you have CFS symptoms
- Add in Prescript Assist after 1-2 weeks – you will keep taking it while you have CFS symptoms
- Add in Bifidobacteria Probiotics after 1-2 weeks — See this post for some possible brands.
- You may (for cost reasons) to rotate thru different brands so you get as many species as you can find.
- Add in MegaSporeBiotic or Just Thrive (for many Bacillus species) after 1 week – at this point you will be taking every Bacillus probiotic available.
- Warning: Some of these are histamine producers. If you are histamine sensitive, use Enterogermina.
- I have a poll to collect actual experiences.
- After 2 weeks, rotate between Mutaflor and Symbioflor-2 every week – you will be taking every E.Coli probiotic available
- After 6 weeks, stop the E.Coli and take the Lactobacillus Gasseri (explained in my next post).
- You want D-Lactic free and H2O2 producing Lactobacillus species/strains
- Lactobacillus usually kill E.Coli so it is one or the other.
- After 6 weeks, stop the Lactobacillus and rotate back to the E.Coli
After 2 Lactobacillus/E.Coli cycles, if there is not significant improvement (or improvement has stopped), then consider a uBiome test. When you get the results, consider using herbs/antibiotics if appropriate. For example, one uBiome results had Rickettsia which would suggest antibiotics.
- Propionibacterium freudenreichii (Securil) is not included because it depends on Lactobacillus helveticus. It is suggested that you eat Emmental cheese instead “An estimated one billion living cells of P. freudenreichii are present in one gram of Emmental.”[Wikipedia]
“Add in” – start with a low dosage (where practical) and increase to recommended dosage on the bottle or higher (2x — to be discussed with your medical professional).
WARNING: Prescript Assist and Prescript Assist Pro are two different products made by two different companies. Only Prescript Assist has had studies on PubMed, the other one is recent to the market and has less variety.
NOTE: This post has been updated by https://atomic-temporary-42474220.wpcomstaging.com/2017/10/08/update-and-recap/ , which is now pinned to the home page.